
The epidemiology of cutaneous malignant melanoma (CMM) continues to evolve [1]. Whilst there has been a small but steady increase in incidence of the tumour there has been a significant decrease in lethality. There are several reasons for this; public health campaigns with an emphasis on prevention, the diagnosis and excision of early-stage disease and the progress in development of new treatments for advanced stage lesions.
The pathogenesis, progression and staging of the disease are related to a number of both intrinsic and extrinsic factors. Of the extrinsic factors one of the most complex and significant is stress. The initiators of the stress response can be both physical and psychological. In the context of this paper, it is important to appreciate that major cosmetic surgical interventions can result in significant surgical stress resulting in a physical stress response. In addition, if the surgery goes wrong and there are significant complications especially when associated with pain, then this can result in a significant psychological stress response. It is imperative that patients who have been diagnosed with a malignant melanoma who are then contemplating cosmetic surgery should be fully informed of the potential risk of disease progression, upstaging of the tumour and reduced chance of five-year survival [2].
Malignant melanoma – definition
A malignant melanoma arises from a malignant transformation of a melanocyte. The melanocytes arise from neural crest cells in the embryo. The neural crest cells migrate widely and give rise to such diverse cell types as the peripheral nerves, Schwann cells, adrenal medulla cells and melanocytes. Melanocytes are pigment-producing cells found mainly in the basal layer of the epidermis of the skin. They synthesise melanin which protects against ultraviolet radiation. In a malignant melanoma these melanocytes undergo a genetic mutation that disrupts normal cellular controls such as growth, differentiation and apoptosis. The result is a clonal proof proliferation of atypical melanocytes with the capacity to invade, to metastasise and to resist immune surveillance.
Epidemiology
Worldwide melanoma diagnoses are in the order of a few hundred thousand new cases per year (GLOBOCAN estimates approximately 330,000 new cases in recent global tallies). Incidence is highest in fair-skinned populations (Australia/New Zealand, North America, Northern and Western Europe) but there is large regional variation [3].
Overall, many high-income countries saw large increases in melanoma through the late 20th and early 21st centuries. In the last decade the picture has become more mixed: some countries (and older age groups) continue to show rising or record-high rates, while others – notably younger age groups in Australia and parts of New Zealand – have shown declines (likely reflecting successful prevention programmes and demographic change). In short: global burden remains substantial, but trends vary strongly by country and age.
Survival/prognosis [4]
Survival has improved a lot. When melanoma is diagnosed early (localised), five-year relative survival is essentially excellent (>99% in recent SEER/US/ACS figures). Regional or distant spread still carries much lower survival (regional approximately 60–75%, distant approximately 30–35%), but new systemic treatments (targeted therapy and modern immunotherapy introduced in the 2010s) have materially improved outcomes for advanced disease. Overall combined five-year relative survival in recent series is in the 90%+ range for many high-income populations. Absolute burden rises with age: older adults (especially those aged 50+) carry the highest absolute incidence and the largest number of cases and deaths in most countries. Many recent increases in case counts reflect ageing populations and higher detection among older cohorts.
Younger cohorts show a complex pattern: historically there were sharp increases in melanoma among younger adults (and especially younger women) in several countries. Recent research finds a mixed picture. Some high-income settings report declines or stabilisation among adolescents and young adults (Australia is a leading example), while other analyses detect rising incidence in certain birth cohorts (for example Generation X in some datasets). Broadly: some younger birth cohorts showed faster rises historically, but trends are heterogeneous by country and are now diverging by generation and place.
The global melanoma burden remains significant (hundreds of thousands of new cases yearly) but trends are heterogeneous: rising in many older age groups and some countries, falling or stabilising among youth where strong prevention has been sustained.
Survival today is much better than two decades ago (early detection and modern therapies are the main drivers) but metastatic melanoma still has substantial mortality. Public health policies (sun protection, reduced tanning-bed use) and early detection remain the most powerful levers to reduce both incidence and mortality.
Aetiology and pathogenesis [5]
There are a number of intrinsic and extrinsic factors which influence or determine the stage and progression of disease.
Intrinsic factors
- Tumour thickness (Breslow thickness)
This is the single most powerful prognostic factor. It is measured from the top of the granular layer of the epidermis to the deepest point of tumour invasion; the deeper the melanoma extends into the dermis the higher the risk of metastases and the lower the percentage five-year survival. - Ulceration
Presence of ulceration over the melanoma surface indicates a more aggressive biology. It reflects rapid tumour growth which outpaces its capacity to generate blood supply and correlates with a poorer prognosis. - Mitotic rate
This is the number of mitoses per square millimetre. It indicates the proliferative activity and tumour aggressiveness. A higher mitotic index predicts a faster progression and a poor prognosis. - Clarke level (anatomical level of invasion)
This describes the anatomical layer reached by the tumour cells:
• Epidermis only (in situ),
• Invades the papillary dermis,
• Fills papillary dermis,
• Reticular dermis,
• Subcutaneous tissues.
The Clarke’s level of invasion has less prognostic significance than the Breslow thickness, but it is still relevant historically. - Genetic and molecular alterations
Specific driver mutations influence behaviour and therapeutic response:
• BRAF mutations promote ma PK pathway activation,
• NRAS and NF1 mutations these are alternate uncle genic roots,
• KIT mutations there are more common in acral and mucosal melanomas.
These mutations determine tumour aggressiveness and guide targeted therapies for example BRAF inhibitors - Tumour microenvironment
This includes stromal cells, immune cells and cytokines. Tumours with robust lymphatic infiltration often respond better to immunotherapy and have improved survival. These are known as hot melanomas. Cold tumours lack immune infiltrations and are more resistant to treatment. - Host immune response and genetics
The ability of the host immune system to recognise and destroy malignant melon melanocytes affects progression. Certain HLA types and immune regulatory gene variance influence prognosis. - Vascular and lymphatic invasion
The presence of tumour cells within vessels signifies early metastatic potential. Acral lentiginous and mucosal melanomas often present later and behave more aggressively. Superficial spreading melanomas usually have better outcome when detected early.
In summary, a malignant melanoma is a melanocyte that has gone genetically rogue acquiring the hallmarks of a cancer: autonomous growth, invasion, metastases and resistance to death signals. The stage and prognosis depend primarily on how deeply the initial tumour has invaded, how rapidly the cells are dividing and its underlying genetic and immune characteristics.
Extrinsic factors
There are number of environmental or external factors that play a significant role in the initiation, progression and prognosis of malignant melanoma. They often interact with intrinsic factors. The following is a list of key factors known to influence disease progression.
- Ultraviolet (UV) radiation exposure
Chronic or intermittent intense exposure to UV, especially UV B, promotes DNA damage, oxidative stress and immunosuppression in the skin.
Continued UV exposure after diagnosis can:
• Promote recurrence or second primary melanomas,
• Enhance tumour aggressiveness via DNA mutation (for example BRAF come up NRAS),
• Impaired local immune surveillance through UV induced immunosuppression. - Immunosuppression (iatrogenic or environmental)
Immunosuppressed states, for example due to organ transplantation, HIV infection or chronic corticosteroid or biological therapy, reduce the host’s ability to detect and destroy malignant cells. This leads to foster tumour growth and metastatic spread in addition to poor treatment response especially to immunotherapy - Lifestyle factors
Smoking is associated with increased oxidative stress and impaired immune response. This may worsen the prognosis. Alcohol consumption suppresses immune function and can alter drug metabolism. Chronic low intake of antioxidants and vitamin D deficiency may impair immune surveillance and DNA repair mechanisms. A sedentary lifestyle is linked to poorer immune function and higher inflammation possibly influencing tumour progression - Infection and inflammation
Chronic inflammation or infection can create a microenvironment conducive to tumour progression. Human papilloma virus (HPV) or herpes virus infections have been proposed as possible cofactors in melanoma progression although the evidence remains limited - Sunbed use (artificial UV)
Artificial tanning devices emit predominantly UV A radiation causing deep dermal DNA damage and immunosuppression, associated with both melanoma initiation and more aggressive disease patterns in younger individuals. - Socioeconomic and health system factors
Delayed diagnosis due to lack of awareness or access to dermatological care can allow disease progression before treatment begins. Cultural differences in sun protection behaviour and screening practices also influence outcomes. - Treatment related factors
Suboptimal surgical margins or incomplete excision can lead to local recurrence. Delayed or inappropriate management therapy affects disease control. Drug interactions or poor compliance can reduce the effectiveness of therapy. - Stress [6]
Both acute and chronic stress can cause hormonal and neuro endocrine cascades that result in immune dysregulation and promotion of inflammatory and angiogenic pathways conducive to tumour progression.
Stress as a pathophysiological concept
Stress represents a systemic physiological and psychological response to perceived threat or challenge, orchestrated primarily through activation of the hypothalamic-pituitary-adrenal (HPA) axis and the sympathetic-adrenomedullary (SAM) system.
Acute stress elicits adaptive ‘fight or flight’ responses – mobilising energy, enhancing alertness and transiently augmenting immune surveillance. However, chronic or unresolved stress leads to maladaptive consequences: sustained cortisol and catecholamine release, immune dysregulation, impaired wound healing and promotion of inflammatory and angiogenic pathways conducive to tumour progression.
In the context of cosmetic surgery, preoperative psychological stress may alter neuroendocrine and immune balance, potentially impairing healing and increasing susceptibility to infection or inflammation. When surgery yields disappointing or disfiguring results, the subsequent emotional stress, characterised by grief, shame, poor body-image, distress, can amplify neuroimmune dysregulation.
Physical stress response [7–10]
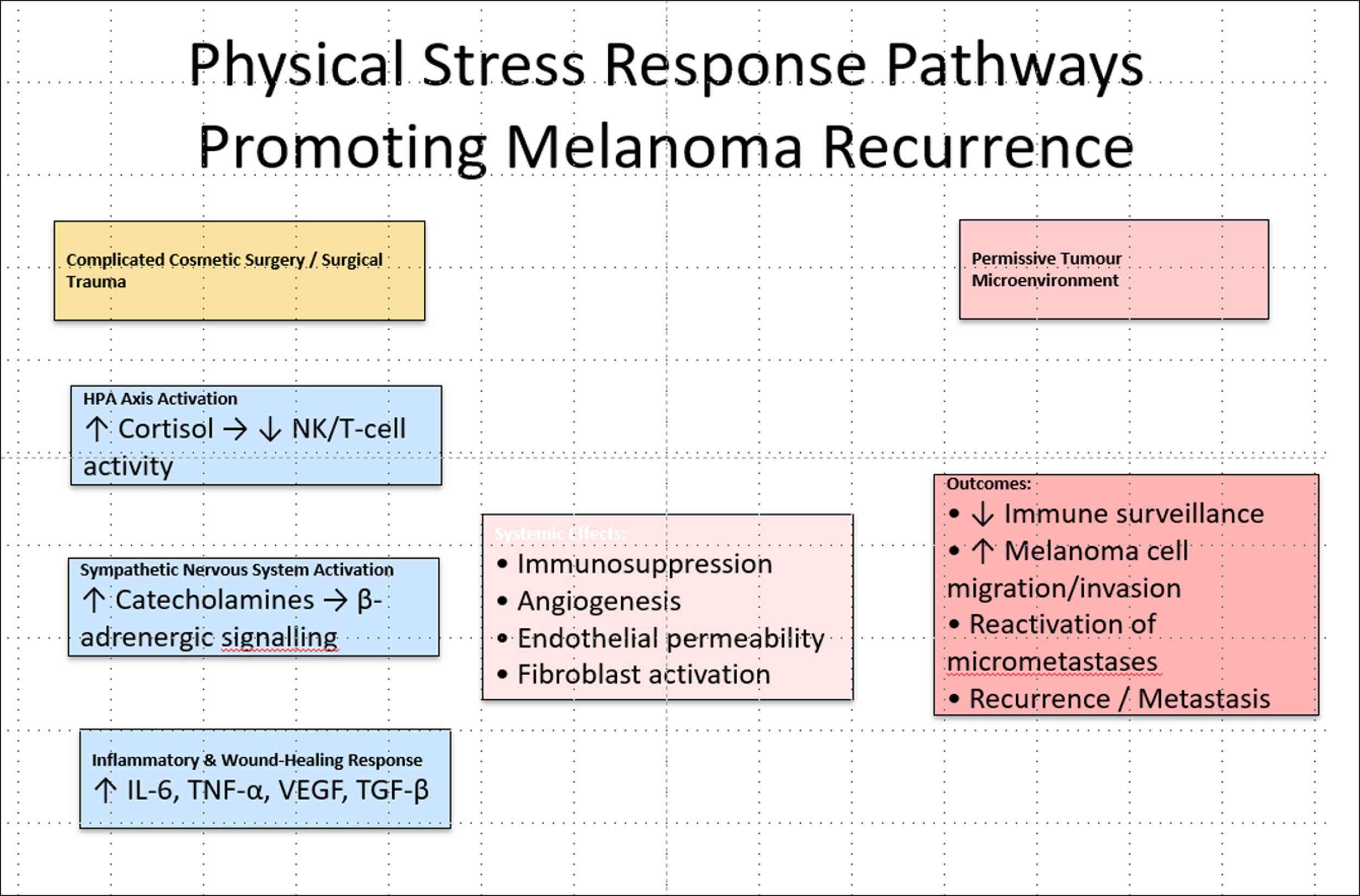
Major or complicated surgery provokes a profound systemic physiological stress response involving activation of the HPA axis, sympathetic nervous system (SNS) and inflammatory cascades. Elevated cortisol and catecholamines suppress cell-mediated immunity that are central to melanoma immunosurveillance (notably natural killer (NK) cell and cytotoxic T-lymphocyte activity). Simultaneously, surgical trauma releases damage-associated molecular patterns (DAMPs) and proinflammatory cytokines (IL-1β, IL-6, TNF-α), promoting angiogenesis, endothelial permeability and melanoma cell extravasation. The perioperative inflammatory milieu, coupled with wound-healing growth factors (VEGF, TGF-β), can reactivate dormant melanoma micrometastases or enhance residual cell proliferation. Physical stress-induced neuroendocrine–immune dysregulation thus creates a transient, but biologically permissive window, for recurrence or metastatic progression following previous melanoma excision.
Severe psychological stress and pain response [12,13].
Severe psychosocial stress following a failed major cosmetic surgery can promote recurrence of a previously excised melanoma through physical neuro-endocrine and inflammatory pathways as well as by psychological mechanisms. Stress-activated HPA-axis elevation of cortisol and chronic sympathetic activation with catecholamine release suppress natural-killer and cytotoxic T-cell surveillance, dysregulate wound-healing cytokines (IL-6, TGF-β, VEGF) and increase angiogenic and invasive signalling via β-adrenergic receptors, together creating a microenvironment permissive to awakening micrometastatic melanoma cells and facilitating invasion. These effects are amplified by stress-related behaviours (sleep loss, alcohol, smoking) and by impaired follow-up/adherence, and may be modifiable (e.g., stress-management interventions or pharmacologic β- adrenergic blockade) as suggested by preclinical and clinical studies
Summary of stress factors
For a patient with malignant melanoma, this combined stress burden may be particularly deleterious. Elevated cortisol and norepinephrine levels have been shown to suppress cytotoxic T-cell and natural killer cell activity, facilitate melanoma cell migration and angiogenesis, and enhance tumour-associated macrophage recruitment. All of these may accelerate disease progression. Thus, psychological and physiological stressors surrounding cosmetic surgery, especially when coupled with profound disappointment, may converge to produce a biopsychoneuroimmunological milieu that fosters melanoma recurrence or metastasis.
Cosmetic surgery
Cosmetic surgery can be defined a surgery that is not medically indicated but sought after by patients who are looking for an enhancement in their body image and sense of wellbeing. In many countries there is no recognised specialty of cosmetic surgery. This is the case in the UK and in Australia and the result is that cosmetic surgery is performed by a range of different doctors. There are specialist surgeons who have been trained in a particular discipline such as plastic surgery or ear nose and throat surgery or dermatological surgery who perform cosmetic surgery as part of their specialist practice. There are other cosmetic surgeons who have no specialist training but have sought procedure-based competencies through personalised training plans.
The procedures
Cosmetic surgery procedures represent a very wide range of interventions. It is possible to categorise these arbitrarily into minor, intermediate and major. The main factors to consider are:
- The amount of tissue trauma,
- The operative time,
- The type and duration of anaesthesia,
- The blood loss and fluid shifts encountered,
- Postoperative pain and inflammation.
Using these factors it's possible to derive a list of cosmetic procedures and categorise them according to their complexity. I have previously published such a list generated by AI and online at https://www.thepmfajournal.com/news/post/part-four-aye-aye-ai [14].
Irrespective of whether a surgeon is trained by specialty or procedure, it is a basic requirement that they are aware of the surgical stress response elicited. Severity categories (minor, intermediate, major) do correlate with surgical stress response, but not perfectly, as factors like operative time, tissue trauma, blood loss and anaesthesia duration play key roles.
It is an unfortunate fact of life that not all surgical procedures progress smoothly and complications do occur. Complications that occur during or after cosmetic surgery can amplify the surgical stress response by prolonging inflammation, increasing metabolic demands and exacerbating systemic physiological disruptions.
Clinical implications
A malignant melanoma is a complex tumour intimately linked to the immune system. When detected early and fully excised, the outcome is excellent as determined by the chance of five-year survival from the disease being over 98%. The tumour does however have the potential to recur locally and to spread to regional lymph nodes and to metastasise widely. Disease progression is related to a complex interaction of both intrinsic and extrinsic factors. Upstaging the disease is associated with a significantly worse prognosis. Factors compromising the effectiveness of the immune system are a particular concern and amongst these is stress. Physical and psychological stress can create a permissive environment that allows a tumour to spread and increase in lethality.
Major cosmetic surgery, particularly when associated with complications, pain and patient dissatisfaction, can result in significant stress that could alter tumour behaviour. There are currently no best practice guidelines that help a cosmetic surgeon counsel a patient who gives a history of prior excision of a skin cancer. There should be [14].
Unfortunately, this is not a theoretical concern. In generality, experts in the management of patients with melanoma are not experts in performing cosmetic surgery whilst experts in cosmetic surgery are not experts in oncology. Clinical practice guidelines can bridge the gap and ensure that clinical decision making is based on what is in the patient’s best interest.
The cosmetic surgeon must confirm the cancer diagnosis. This is done through the process of taking a mandatory previous medical history from the patient. If they declare that they have been treated for ‘skin cancer’ then it is imperative for the cosmetic surgeon to clarify the type of skin cancer, where it was located and when it was removed. Patients who have had a malignant melanoma removed should be under the care of an oncologist. The cosmetic surgeon must liaise with the oncologist to confirm the histological analysis of the tumour and the stage of the disease.
Clinical practice guidelines need to be developed by the relevant stakeholders to determine the appropriate interval between melanoma diagnosis and cosmetic surgical intervention, based on the percentage chance of disease recurrence. Table 1 features a suggestion based on contemporary figures:
Table 1: Melanoma stage and recurrence risk [15]:
-
Thin melanoma (Stage IA–IB, ≤1mm thickness, no ulceration): lower recurrence risk, but follow-up for at least one to two years is often recommended before elective cosmetic procedures.
-
Intermediate-thickness melanoma (Stage II, 1–4mm thickness): higher recurrence risk, requiring at least two to three years of disease-free survival before elective surgery.
-
Advanced melanoma (Stage III–IV, nodal or metastatic involvement): high recurrence risk; elective surgery is typically discouraged unless disease-free for at least five years.
The cosmetic surgeon then must discuss with the oncologist and the patient the potential risk of the proposed operation regarding the magnitude of the surgical stress and the possibility of complications adding to the stress. As pathological stress levels can lead to immune dysfunction and disease progression the risk of upstaging the melanoma must be clearly discussed with the patient before they can give a fully informed consent to the procedure.
If a patient does consent to a procedure, then it is important for the cosmetic surgeon to appreciate the relevance of oncological follow-up and surveillance. This relates to the importance of examination for local, in transit and regional node recurrence. This means that the tissues around the site of the primary excision should not be disturbed by additional scarring or inflammation.
This paper presents the justification for developing best/clinical practice guidelines for cosmetic surgeons contemplating a major cosmetic procedure in a patient with a recent history of excision of a cutaneous malignant melanoma. The details need to be addressed by the stakeholders, but the guidelines should cover:
- Confirm the diagnosis of cutaneous malignant melanoma.
- Liaise with the oncologist.
- Allow a disease-free period before operating based on the stage of the tumour.
- Fully inform the patient of the potential risks of upstaging the tumour
- Ensure that any cosmetic procedure will not interfere with oncological disease surveillance.
References
1. Waseh S, Lee JB. Advances in melanoma: epidemiology, diagnosis, and prognosis. Front Med 2023;10:1268479.
2. De Pinto G, Mignozzi S, La Vecchia C, et al. Global trends in cutaneous malignant melanoma incidence and mortality. Melanoma Res 2024;34(3):265–75.
3. Arnold M, Singh D, Laversanne M, et al. Global burden of cutaneous melanoma in 2020 and projections to 2040. JAMA Dermatol 2022;158(5):495–503.
4. Caraviello C, Nazzaro G, Tavoletti G, et al. Melanoma skin cancer: a comprehensive review of current knowledge. Cancers 2025;17(17):2920.
5. Centeno PP, Pavet V, Marais R. The journey from melanocytes to melanoma. Nat Rev Cancer 2023;23(6):372–90.
6. Switzer B, Puzanov I, Gandhi S, Repasky EA. Targeting beta-adrenergic receptor pathways in melanoma: how stress modulates oncogenic immunity. Melanoma Res 2024;34(2):89–95.
7. Ben-Eliyahu S. The promotion of tumour metastasis by surgery and stress: neuroendocrine mechanisms. Brain Behav Immun 2003;17 Suppl 1:S27–S36.
8. Hildenborg M. Systemic inflammation and neuroimmunity in surgical trauma. Karolinska Institutet (Thesis); 2024.
9. Neeman E, Ben-Eliyahu S. Surgery and stress promote cancer metastasis: new outlooks on perioperative mediating mechanisms and immune involvement. Brain Behav Immun 2013;30(Suppl):S32–40.
10. Cole SW, Sood AK. Molecular pathways: Beta-adrenergic signaling in cancer. Clin Cancer Res 2012;18(5):1201–06.
11. De Giorgi V, Grazzini M, Benemei S, et al. Propranolol for off-label treatment of patients with melanoma. JAMA Oncol 2018;4(2):e172908.
12. Antoni MH, Lutgendorf SK, Cole SW, et al. The influence of bio-behavioural factors on tumour biology: pathways and mechanisms. Nat Rev Cancer 2006;6(3):240–8.
13. De Giorgi V, Geppetti P, Lupi C, Benemei S. The role of β-blockers in melanoma. J Neuroimmune Pharmacol 2020;15(1):17–26.
14. https://www.thepmfajournal.com/
news/post/part-three-aye-aye-ai
15. Papageorgiou C, Apalla Z, Manoli SM, et al. Melanoma: staging and follow-up. Dermatol Pract Concept 2021;11(Suppl 1):e2021162S.
This list of references is not an exhaustive list but a representative sample of relevant papers.
Declaration of competing interests: None declared.






