Botulinum toxin type A (BoNT-A) is a potent neurotoxin produced by the bacterium Clostridium botulinum [1]. BoNT-A has been used for various therapeutic and aesthetic purposes, including the treatment of muscle disorders, chronic pain and facial wrinkles [2-4]. Over the years, multiple BoNT-A products have been developed and marketed with varying degrees of safety and efficacy. All these products have differing manufacturing processes and are hence not interchangeable.
This article aims to briefly review the development and evolution of BoNT-A products, comparing their safety and efficacy in various therapeutic and aesthetic applications. The discussion spans from the early days of BoNT-A to the recent advances and emerging products in the market. Emphasis is placed on the differences in potency, formulation, and clinical applications of these products.
History of botulinum toxin type A
The history of BoNT-A dates back to the early 19th century. It has since evolved from a deadly toxin to a versatile therapeutic agent used in various medical and cosmetic applications.
Discovery (1817-1895): The story of botulinum toxin begins in 1817 when German Physician Justinus Kerner first described the clinical features of foodborne botulism, a severe paralytic illness caused by ingestion of the toxin produced by the bacterium Clostridium botulinum [5]. In 1895, Belgian Microbiologist Emile van Ermengem isolated and identified the causative organism, Clostridium botulinum, from a contaminated ham that caused a botulism outbreak [6].
Purification and classification (1920s-1940s): The first purified preparation of botulinum toxin was achieved in the 1920s by Hermann Sommer and colleagues at the University of California, San Francisco [7]. Further research in the 1940s led to the classification of botulinum toxin into seven distinct serotypes (A-G) based on their antigenic properties [7,8].
Therapeutic potential (1950s-1970s): In the 1950s, researchers began to explore the therapeutic potential of botulinum toxin. Vernon Brooks discovered that BoNT-A could selectively block the release of acetylcholine at neuromuscular junctions, causing temporary muscle paralysis [9]. This discovery laid the foundation for the development of BoNT-A as a therapeutic agent.
Clinical use (1980s-present): In 1989, the US Food and Drug Administration (FDA) approved onabotulinumtoxinA (Botox®) for the treatment of strabismus and blepharospasm [10]. Since then, the clinical use of BoNT-A has expanded to include various medical and cosmetic applications, such as the treatment of cervical dystonia, chronic migraine, overactive bladder, and facial wrinkles [3,11].
New formulations and future developments: In recent years several BoNT-A formulations have been developed, each with unique properties and characteristics. Ongoing research aims to improve the safety and efficacy of these products and explore new therapeutic applications [12,13]. With the ongoing research and introduction of new formulations, the therapeutic potential of BoNT-A continues to expand, providing effective treatment options for a wide range of medical and cosmetic applications. As understanding of this versatile neurotoxin grows, it is likely that we will continue to witness new applications and improvements in the safety and efficacy of BoNT-A products.
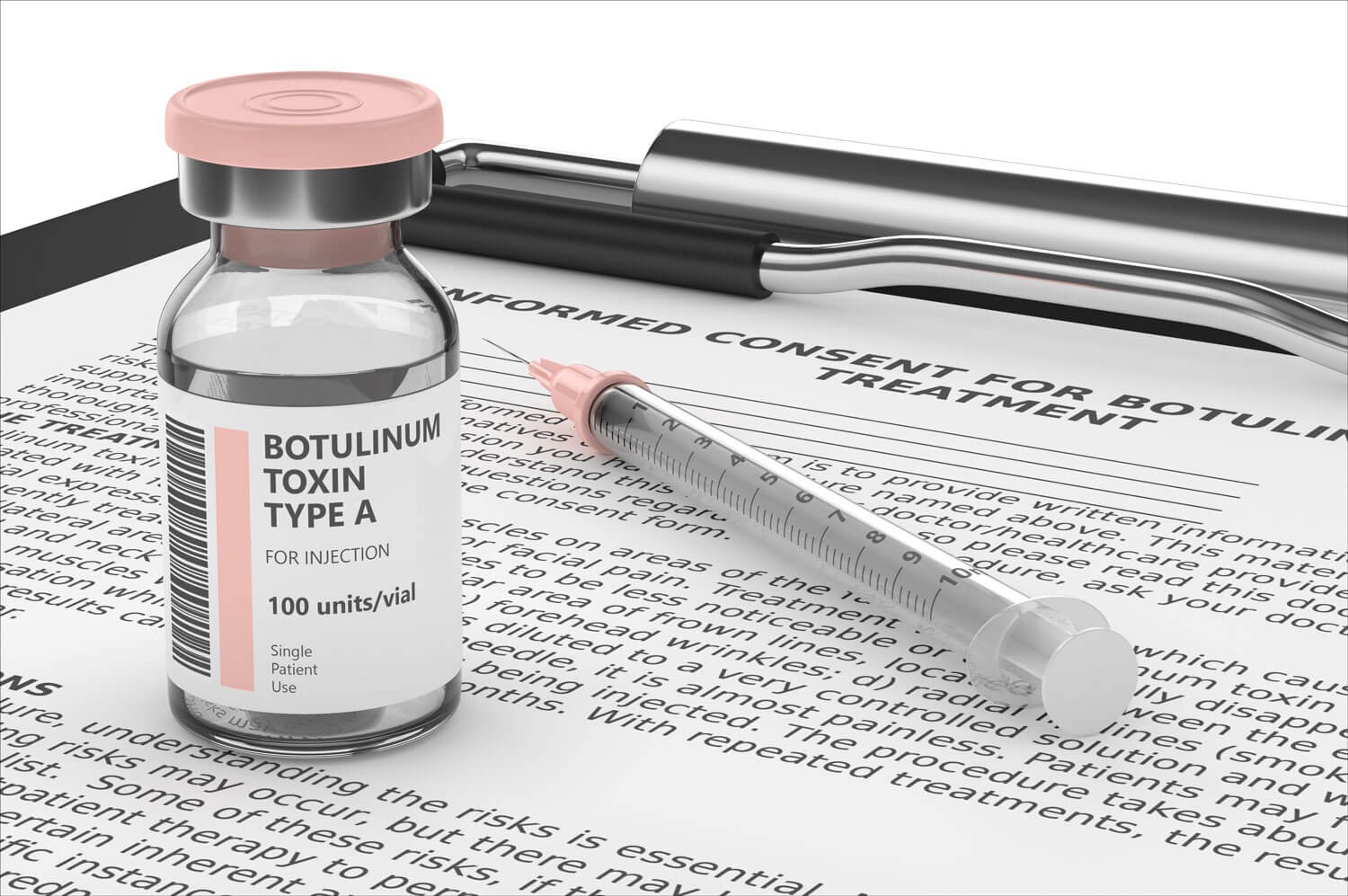
The various BoNT-A formulations
OnabotulinumtoxinA (Botox® / Vistabel®): This is a vacuum-dried powder formulation of BoNT-A produced by Allergan Inc. (Dublin, Ireland). Botox has been widely used for therapeutic and aesthetic purposes for many years, with its safety and efficacy well-established [2,14].
AbobotulinumtoxinA (Dysport® / Azzalure®): This is a lyophilised powder formulation of BoNT-A developed by Ipsen (Paris, France) and Galderma (Lausanne, Switzerland). Dysport has demonstrated safety and efficacy comparable to OnabotulinumtoxinA in various studies, although it exhibits different diffusion / spread properties, potentially leading to different clinical outcomes [15,16].
AbobotulinumtoxinA injectable solution (Alluzience®): This is a liquid form of botulinum toxin type A (Galderma, Lausanne, Switzerland). Owing to its liquid state, it offers the possibility of enhanced safety and dosing precision in comparison to freeze-dried BoNT-A formulations.
IncobotulinumtoxinA (Xeomin® / Bocouture® / NT 201): This lyophilised powder formulation of BoNT-A is produced by Merz Pharmaceuticals GmbH (Frankfurt, Germany). Xeomin contains a purified neurotoxin without accessory proteins, potentially reducing antigenicity and adverse reactions [17]. Seo et al. compared the safety and efficacy of Xeomin with Botox and Dysport, finding it to be effective, but with differences in potency, immunogenicity, and diffusion / spread [18].
PrabotulinumtoxinA (Nuceiva® / Jeuveau®): Developed by Daewoong Pharmaceutical Co., Ltd. (Seoul, South Korea), this lyophilised powder formulation of BoNT-A aims to improve upon the safety and efficacy profiles of its predecessors. In 2018 Dhillon et al. discussed the potential advantages of PrabotulinumtoxinA in specific clinical applications due to its field of action [13], while in 2020 another study compared reported safe, effective and non-inferior results for PrabotulinumtoxinA in comparison to OnabotulinumtoxinA for the treatment of moderate to severe glabellar lines in adults [19]. Other studies have also reported on the safety and efficacy of PrabotulinumtoxinA [20-22].
DaxibotulinumtoxinA (DAXXIFYTM, RT002): This lyophilised powder formulation of BoNT-A is produced by Revance Therapeutics, Inc. (USA). It is a highly purified 150-kDa core neurotoxin derived from the Hall strain of Clostridium botulinum. Several studies have reported high efficacy and safety rates along with good tolerance levels for DaxibotulinumtoxinA [23-25].
LetibotulinumtoxinA (Botulax / Hutox and Letybo): Both of these lyophilised powder formulations of BoNT-A are produced by Hugel Pharma (Korea). Croma Pharma has licensed the product from Hugel Inc. (Letybo). Their safety and efficacy profiles are currently being evaluated with one study demonstrating high efficacy and a convincing safety profile in the treatment of glabellar lines [26].
Neuronox and Innotox: These lyophilised powder and liquid formulations of BoNT-A are produced by Medytox (Korea). Research on these products is ongoing, with some studies showing promising safety and efficacy results in various therapeutic and aesthetic applications [27,28].
Duration of effect of different BoNT-A formulations
The duration of effect of BoNT-A formulations varies depending on the specific product, the treated condition, the injected dose, and individual patient factors. Here, we discuss the duration of effect for some of the most commonly used BoNT-A formulations:
OnabotulinumtoxinA (Botox® / Vistabel®): The duration of effect for Botox® generally ranges between three to six months, depending on the treated condition and the injected dose. In cosmetic applications, such as the treatment of facial wrinkles, the effect typically lasts for about three to four months [29,30]. For medical conditions like cervical dystonia or chronic migraine, the duration of effect may extend up to six months [31,32].
AbobotulinumtoxinA (Dysport® / Azzalure®): Similar to Botox, the duration of effect for Dysport® typically ranges from three to six months. In cosmetic applications, the effect lasts for approximately three to four months [33]. For medical conditions the duration of effect can extend up to four to six months [11].
IncobotulinumtoxinA (Xeomin® / Bocouture® / NT 201): The duration of effect for Xeomin is comparable to that of Botox and Dysport. In cosmetic applications, the effect generally lasts for about three to four months [34]. In the treatment of medical conditions the duration of effect can last for up to six months [35].
PrabotulinumtoxinA (Nuceiva® / Jeuveau®): The duration of effect for Nuceiva appears to be similar to that of other BoNT-A formulations, with effects lasting for approximately three to four months in cosmetic applications [36].
DaxibotulinumtoxinA (DAXXIFYTM, RT002): DaxibotulinumtoxinA has shown promise in providing a longer duration of effect compared to other BoNT-A products. In a phase three clinical trial for glabellar lines, the median duration of effect was found to be approximately 24 weeks so roughly six months [37].
As newer formulations of BoNT-A are being developed, their duration of effect and efficacy in various applications may differ from those of the more established products. Research is ongoing, and additional clinical studies may provide further insights into the comparative performance of these formulations.
It is essential to consider that the duration of effect for BoNT-A formulations can vary among individuals due to factors such as age, muscle mass, and metabolism. Furthermore, the injected dose, dilution, and injection technique can also influence the duration of effect. Clinicians should tailor treatment plans based on individual patient needs and the specific indications for which BoNT-A is being used. Emerging formulations show promise in providing a longer duration of effect, which may expand the range of applications and improve patient satisfaction with treatment outcomes.
Immunogenicity and BoNT-A formulations
Immunogenicity refers to the ability of a substance to provoke an immune response in the body. Immunogenicity is an important consideration in the selection of BoNT-A formulations for clinical use.
With relation to BoNT-A injections, immunogenicity can lead to the development of neutralising antibodies against the neurotoxin, potentially reducing its therapeutic effectiveness and increasing the risk of treatment failure [38,39].
Different BoNT-A formulations exhibit varying degrees of immunogenicity due to differences in their composition, including hypotheses regarding the presence or absence of complexing proteins and other impurities [17,40]. The use of lower doses and less frequent injections might help minimise the risk of antibody formation [38].
Conclusion
BoNT-A products have evolved significantly since their initial introduction for therapeutic and aesthetic applications. While older formulations like Botox®, Dysport® and Xeomin® have demonstrated proven safety and efficacy, newer products and emerging formulations from Korea and China are aiming to improve upon these profiles. The products are all unique and are not interchangeable. Differences in potency, immunogenicity, field of effect and spread / diffusion / migration impact the clinical performance of all formulations. Continued research is essential to optimise the safety and efficacy of BoNT-A products, ensuring they meet the diverse needs of patients and clinicians.
References
1. Schantz EJ, Johnson EA. Properties and use of botulinum toxin and other microbial neurotoxins in medicine. Microbiological Reviews 1997;61(1):80-99.
2. Carruthers JD, Carruthers A. Treatment of glabellar frown lines with C. botulinum-A exotoxin. Journal of Dermatologic Surgery and Oncology 1992;18(1):17-21.
3. Dressler D, Saberi FA. Botulinum toxin: mechanisms of action. European Neurology 2005;53(1):3-9.
4. Jabbari B. Botulinum neurotoxins in the treatment of refractory pain. Nature Reviews Neurology 2013;9(12):676-88.
5. Kerner J. Neue Beobachtungen auf dem Gebiete der Gifte: Wirkung des Wurstgiftes auf den thierischen Organismus. Tübingen: Osiander. 1817.
6. Van Ermengem E. Ueber einen neuen anaëroben Bacillus und seine Beziehungen zum Botulismus. Zeitschrift für Hygiene und Infektionskrankheiten 1897;26(1):1-56.
7. Lamanna C. The most poisonous poison. Science 1946;104(2699):419-22.
8. Burke GS. Immunological relationships of strains of Clostridium botulinum. Journal of Bacteriology 1949;58(4):491-6.
9. Brooks VB. The action of botulinum toxin on the neuro-muscular junction. The Journal of Physiology 1953;121(3):579-92.
10. Scott AB. Botulinum toxin injection into extraocular muscles as an alternative to strabismus surgery. Journal of Pediatric Ophthalmology and Strabismus 1989;26(5):221-4.
11. Truong D, Brodsky M, Lew M, et al. Long-term efficacy and safety of botulinum toxin type A (Dysport) in cervical dystonia. Parkinsonism & Related Disorders 2010;16(5):316-23.
12. Frevert J. Content of botulinum neurotoxin in Botox®/Vistabel®, Dysport®/Azzalure®, and Xeomin®/Bocouture®. Drugs in R&D 2015;15(3):285-90.
13. Dhillon S, Do DV, Furuichi M, et al. PrabotulinumtoxinA: A new botulinum toxin type A. Drugs of Today 2018;54(12):727-32.
14. Binder WJ, Brin MF, Blitzer A, Pogoda JM. Botulinum toxin type A (Botox) for treatment of migraine headaches: an open-label study. Otolaryngology-Head and Neck Surgery 2002;127(6):502-7.
15. Odergren T, Hjaltason H, Kaakkola S, et al. A double blind, randomised, parallel group study to investigate the dose equivalence of Dysport® and Botox® in the treatment of cervical dystonia. Journal of Neurology, Neurosurgery & Psychiatry 1998;64(1):6-12.
16. Rystedt A, Zetterberg L, Burman J, et al. A double-blind, randomized, parallel-group study to investigate the dose equivalence of Dysport and Botox in the treatment of hemifacial spasm. Movement Disorders 2002;17(5):1065-9.
17. Frevert J, Dressler D. Complexing proteins in botulinum toxin type A drugs: a help or a hindrance? Biologics: Targets & Therapy 2015;9:325-32.
18. Seo K, Kim J, Choi Y. Comparison of the efficacy and safety of three formulations of botulinum toxin A (Botox®, Xeomin®, and Dysport®) in the management of benign essential blepharospasm: a randomized, double-blind, non-inferiority study. Graefe’s Archive for Clinical and Experimental Ophthalmology 2015;253(7):1065-71.
19. Rzany B-J, Ascher B, Avelar RL, et al. A multicenter, randomized, double-blind, placebo-controlled, single-dose, phase III, non-inferiority study comparing PrabotulinumtoxinA and OnabotulinumtoxinA for the treatment of moderate to severe glabellar lines in adult patients. Aesthetic Surgery Journal 2020;40(4):413-29.
20. Suh Y, Jeong GJ, Noh HJ, et al. A multicenter, randomized, open‐label comparative study of prabotulinumtoxinA with two different dosages and diverse proportional injection styles for the reduction of gastrocnemius muscle hypertrophy in Asian women. Dermatologic Therapy 2019;32(5):e13009.
21. Beer KR, Shamban AT, Avelar RL, et al. Efficacy and Safety of PrabotulinumtoxinA for the Treatment of Glabellar Lines in Adult Subjects: Results From 2 Identical Phase III Studies. Dermatol Surg 2019;45(11):1381-93.
22. Lorenc ZP, Adelglass JM, Avelar RL, et al. The second of two one-year, multicenter, open-label, repeat-dose, phase II safety studies of PrabotulinumtoxinA for the treatment of moderate to severe glabellar lines in adult patients. Aesthetic Surgery Journal 2021;41(12):1423-38.
23. Bertucci V, Solish N, Kaufman-Janette J, et al. DaxibotulinumtoxinA for Injection has a prolonged duration of response in the treatment of glabellar lines: pooled data from two multicenter, randomized, double-blind, placebo-controlled, phase 3 studies (SAKURA 1 and SAKURA 2). Journal of the American Academy of Dermatology 2020;82(4):838-845.
24. Fabi SG, Cohen JL, Green LJ, et al. DaxibotulinumtoxinA for Injection for the treatment of glabellar lines: efficacy results from SAKURA 3, a large, open-label, phase 3 safety study. Dermatologic Surgery 2021;47(1):48.
25. Green JB, Mariwalla K, Coleman K, et al. A large, open-label, phase 3 safety study of DaxibotulinumtoxinA for Injection in glabellar lines: a focus on safety from the SAKURA 3 study. Dermatologic Surgery 2021;47(1):42.
26. Mueller DS, Prinz V, Adelglass J, et al. Efficacy and Safety of Letibotulinumtoxin A in the Treatment of Glabellar Lines: A Randomized, Double-Blind, Multicenter, Placebo-Controlled Phase 3 Study. Aesthet Surg J 2022;42(6):677-88.
27. Kwon HJ, Kim YJ, Choi YJ, Choi YS. Comparison of efficacy and safety between botulinum toxin type A (Botox) and type B (Medytox). Journal of Cosmetic and Laser Therapy 2013;15(6):340-6.
28. Ryu WH, Kim JH, Kim YC, Kang HY. Comparison of two different formulations of botulinum toxin type A for the treatment of glabellar lines: a double-blind, randomized study. Journal of Cosmetic and Laser Therapy 2015;17(5):268-71.
29. Carruthers A, Carruthers J. Botulinum toxin type A treatment of multiple upper facial sites: patient-reported outcomes. Dermatologic Surgery 1998;34(1):15-20.
30. Carruthers JA, Lowe NJ, Menter MA, et al. & BOTOX® Glabellar Lines I, I. I. S. G. A multicenter, double-blind, randomized, placebo-controlled study of the efficacy and safety of botulinum toxin type A in the treatment of glabellar lines. Journal of the American Academy of Dermatology 2002;46(6):840-9.
31. Brin MF, Lew MF, Adler CH, et al. Safety and efficacy of NeuroBloc (botulinum toxin type B) in type A-responsive cervical dystonia. Neurology 1999;53(7):1439-46.
32. Aurora SK, Dodick DW, Turkel CC, et al. OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 1 trial. Cephalalgia 2011;31(7):793-803.
33. Rzany B, Ascher B, Monheit G, European D. Treatment of glabellar lines with botulinum toxin type A (Speywood Unit): a clinical overview. Journal of the European Academy of Dermatology and Venereology 2011;25(1):1-14.
34. Sattler G, Callander MJ, Grablowitz D, et al. Noninferiority of incobotulinumtoxinA, free from complexing proteins, compared with another botulinum toxin type A in the treatment of glabellar lines. Dermatologic Surgery 2010;36(s2):2146-54.
35. Benecke R, Jost WH, Kanovsky P, et al. A new botulinum toxin type A free of complexing proteins for treatment of cervical dystonia. Neurology 2005;64(11):1949-51.
36. Kwon HB, Kim JH, Kim YJ, Park JW. Comparison of the efficacy and safety of prabotulinumtoxinA and onabotulinumtoxinA for lower facial contouring. Plastic and Reconstructive Surgery 2019;144(4):665e-73e.
37. Fabrizio JA, Gupta S, Hadeed J, Carruthers A. DaxibotulinumtoxinA for the treatment of glabellar lines: Results from each of two multicenter, randomized, double-blind, placebo-controlled, phase 3 studies (SAKURA 1 and SAKURA 2). Aesthetic Surgery Journal 2021;41(9):1019-28.
38. Dressler D, Hallett M.) Immunological aspects of Botox®, Dysport® and Myobloc™/NeuroBloc®. European Journal of Neurology 2006;13(s1):11-5.
39. Brin MF, Comella CL, Jankovic J, et al. Long-term treatment with botulinum toxin type A in cervical dystonia has low immunogenicity by mouse protection assay. Movement Disorders 2008;23(10):1353-60.
40. Dressler D, Bigalke H. Botulinum toxin type B de novo therapy of cervical dystonia: frequency of antibody induced therapy failure. Journal of Neurology 2005;252(7):904-7.
Declaration of competing interests: None declared.
COMMENTS ARE WELCOME