With skin cancer now at epidemic levels, early diagnosis is essential. Specialist Stephen Hayes advocates the use of dermoscopy as a triage tool and explains how to interpret the data.
Melanoma skin cancer is now the UK’s fifth most common cancer and killed 2459 people in 2014 [1]. Early detection radically improves outcome. Dermoscopy (or dermatoscopy) is a well-established technique, used worldwide in the diagnosis of skin lesions. There is strong evidence for its effectiveness as a triage tool, screening out benign lesions and enabling earlier diagnosis of melanomas [2]. Given the skin cancer epidemic and limited healthcare resources, dermoscopy by a suitably trained individual should be readily available to all patients worried about a skin lesion.
What does dermoscopy do?
Essentially, dermoscopes illuminate, magnify (x10) and break down refraction, enabling the viewer to see sub-macroscopic structures in the epidermis and top 1 or 2 millimetres of dermis. Details and features that were previously obscure or invisible are seen. As with stethoscope or endoscope, the dermoscope gives us more data to work with. Correct interpretation of this data enables more specific and sensitive diagnosis.
Brief history of dermoscopy
Skin surface microscopy was first used by Kolhaus in Germany in 1663 to examine vessels in the nail matrix. It was refined by the addition of oil immersion by Ernst Abbe in 1878. Early devices were bulky, the first portable dermoscope was introduced in 1958. In 1981 the first paper was published on using the technique to differentiate between benign and malignant melanocytic skin lesions, based on the analysis of pigment network. In 1989, Peter H Soyer published research correlating dermoscopic features with histopathology. In 2003, the International Dermoscopy Society was founded in Graz, Austria, since then the amount of published literature has increased exponentially.
“Melanoma skin cancer is now the UK’s fifth most common cancer. “
Overview of global dermoscopy
At the 2015 World Dermoscopy Congress in Vienna, some 1300 dermatology doctors from 66 nations heard the latest evidence from leading international experts and heard progress reports from Holland, Poland, Chile, India, USA, Russia and many other countries. A Russian delegate told how teledermoscopy had been embraced, with experts in Moscow web-linked to regional centres to give quick diagnoses to doctors in the regions. In South America dermoscopy was being used to evaluate parasitic and inflammatory skin conditions, in Holland GPs were being trained. The Polish delegation told us how they were tackling their appalling melanoma survival rate of 50% by leafleting all health professionals and providing dermoscopy training with help from Viennese expert Professor Harald Kittler.
There were few British delegates and no presentations or posters by UK or Irish doctors. For some reason, the UK has lagged behind in dermoscopy research and implementation. A somewhat larger British contingent is expected at the forthcoming 5th World Congress in Thessaloniki, Greece this 14-16 June [3].
What types of dermoscope exist?
Scopes can be glass / gel contact, no-touch, or a combination of both (hybrid). With contact scopes, a clear viscous contact medium is used to break down the refraction. Liquid paraffin or vegetable oil have been used, but alcohol gel is convenient. A bead of gel is enough, don’t let it run into the patient’s eyes. A wheel adjusts focus, like binoculars. No-touch scopes are held a few centimetres out and moved to and fro between the patient’s skin and the operator’s eye until features come into focus.
Each type has pros and cons. No-touch is quicker and cleaner, good for scanning many lesions fast, but gives a less clear view of deeper structures and is little use for photography. Some dermoscopic structures, like blood vessels and milia-like cysts, are better seen with one mode or the other. Experienced users generally like to have both modes available. Modern hybrid devices, which I favour, can be used in either mode. I use the no-touch for scanning pigmented lesions (most of which are benign) and then deploy the glass / gel plate for closer examination and photography of questionable lesions.
Dermoscopic features of skin lesions
Dermoscopy can confirm most lesions as benign in seconds at the first consultation, saving time, cash and worry. Before starting dermoscopy the clinician must understand the natural history of common and important skin lesions, take a history, inspect and palpate. Having done this, dermoscopy will improve specificity and confidence. It takes time to acquire expertise, but much can be achieved with as little as one day’s training [4].
Dermoscopy has its own language: the beginner must learn how to recognise, name and interpret dermoscopic features of lesions, and set them in context. Books, online resources, courses and sitting in on clinics will help develop this new vocabulary, grammar and comprehension.
Figure 1: Performing dermoscopy.
Figure 2: A bland looking solar lentigo.
Figure 3: Haemangioma with purple clods.
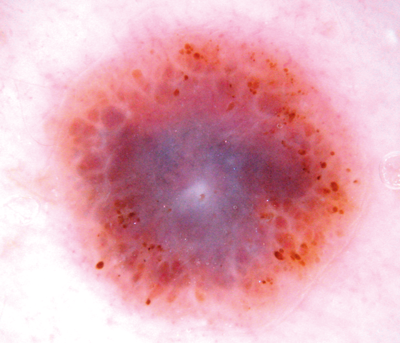
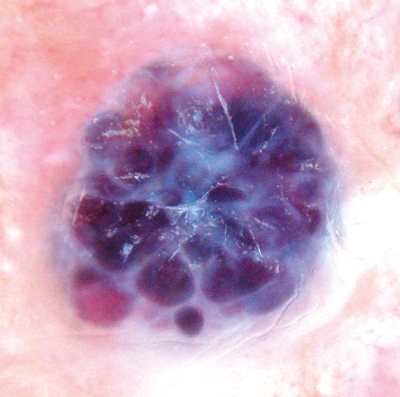
Figure 4: Melanoma with multiple colours, blue veil and atypical globules.
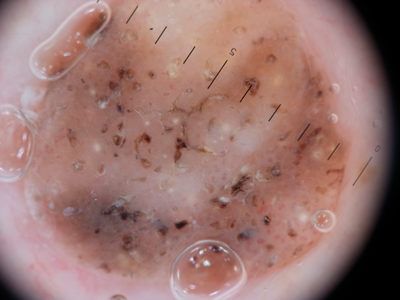
Figure 5: Seborrhoeic wart with fissures, keratin pits and cysts and stuck-on border.
A
B
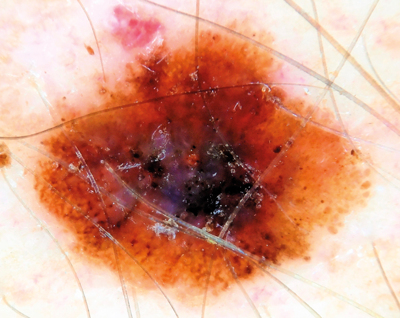
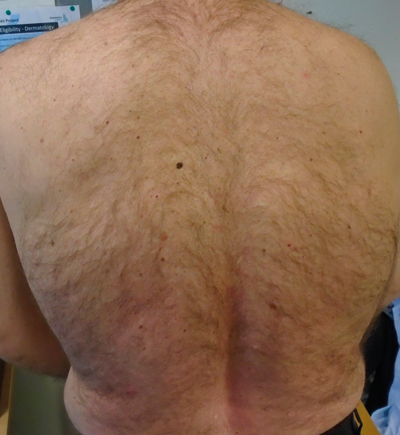
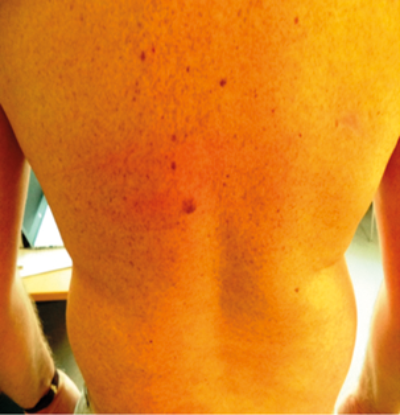
Figure 6 (A and B above): Ugly duckling mole on back. Dermoscopy shows
three colours plus irregular dots and globules. Thin melanoma.
A
B
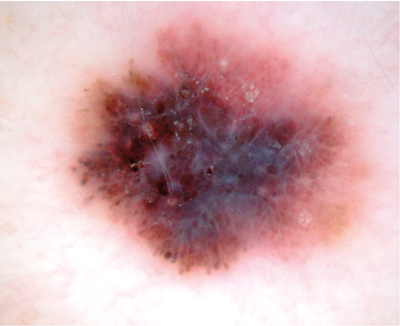
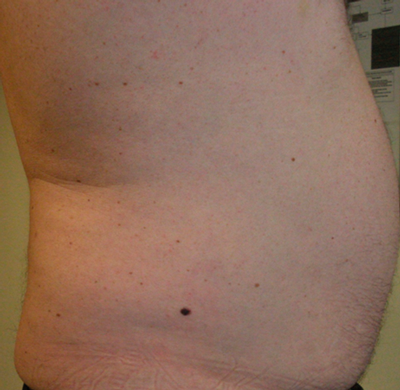
Figure 7 (A and B above): This relatively innocuous mole on dermoscopy shows chaotic colours
and structures including blue grey veil, shiny white streaks and irregular globules = melanoma.
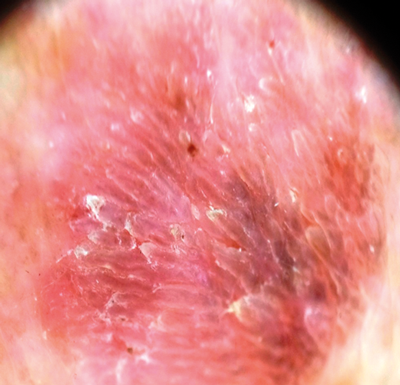
A
A
Figure 8 (A and B above): This pigmented lesion caused concern,
but dermoscopy shows an even papillary pattern = keratosis.
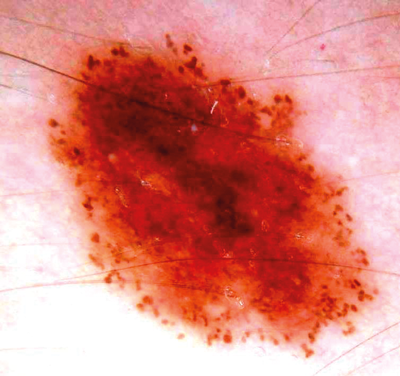
Figure 9: Normal naevus in young person with good
symmetry and regular globules round periphery.

Figure 10: Grossly asymmetrical mole with irregular globules and blue grey veil = melanoma.
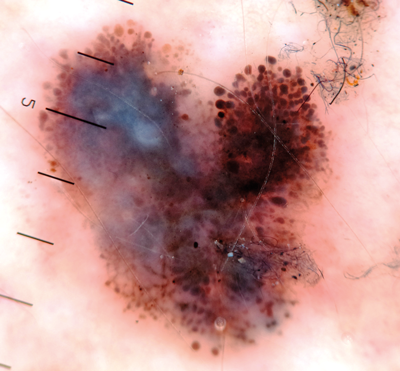
Figure 11: Another melanoma irregular globules and blue white veil.
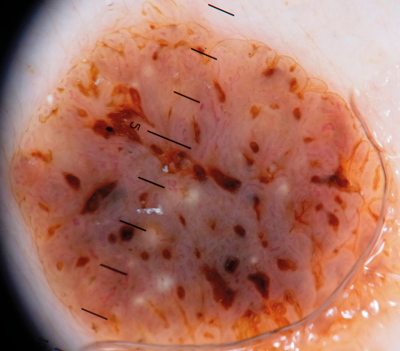
Figure 12: Typical seborrhoiec keratosis with comedo-like openings, milia like cysts, fissures
and a well-defined ‘stuck on’ border. Dermoscopy is great for triaging these lesions out.
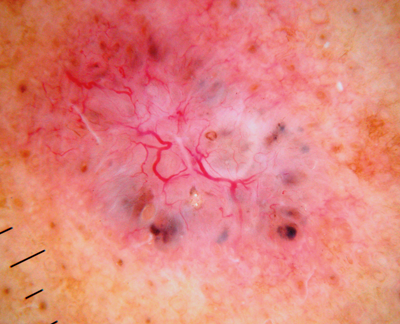
Figure 13: A basal cell cancer with typical arborising vessels, blue clods and some micro erosions.
Dermoscopy can confirm diagnosis, avoiding the need for biopsy, and help define excision margins.
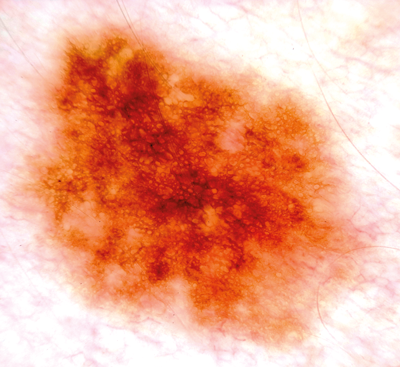
Figure 14: This benign junctional naevus shows one colour (brown)
and one pattern (reticular network) so is harmless.
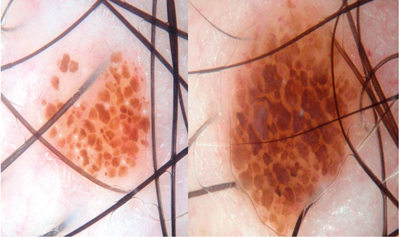
Figure 15: Two cobblestone naevi – one colour, one pattern, harmless.
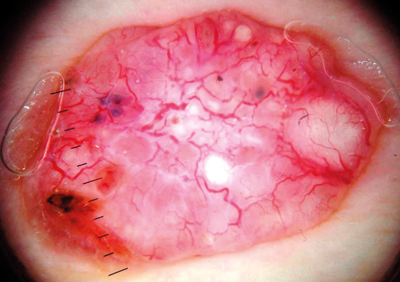
Figure 16: This feature rich BCC shows arborising vessels, blue clods and micro ulcers.
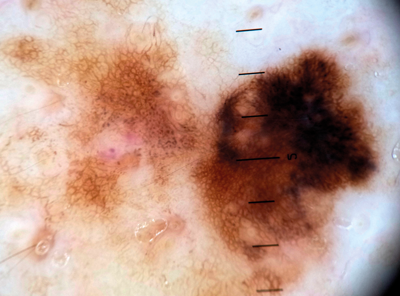
Figure 17: A thin melanoma with atypical network coalescing into an eccentric black blotch.
Dermoscopic features of melanocytic naevi
Flat, mature moles may have a reticular pattern or be featureless (as in blue naevi) or have globules, which represent nests of melanocytes. Where these structures are homogeneous in a stable mole, we need not worry. Globules are seen in normally growing naevi, often in a ring around the periphery of the mole, so an even pattern of globules in a symmetrical, brown naevus in a youngster is fine. However, if the globules are asymmetric in colour, size and distribution, especially with a history of visible change, and if the mole is unique, excision or referral should be considered.
Moles change over time, with globules usually giving way to reticular network past the age of 40. Naevi may become featureless and fade away in older age. The beginner should scope as many banal naevi as possible in patients of all ages to gain a feel for the range of normal. As a rule, if a stable, flat naevus has only one colour and one pattern, it is harmless. Some benign naevi have hypo- or hyperpigmented centres, others have featureless centres and peripheral reticular network, or else multifocal hypopigmentation. Differing shades of brown in itself is no concern – ‘multiple colours’ means not light brown plus medium brown, but two or more of brown, black, blue, red, pink, grey or white.
Check the skin, not just the mole
A patient’s naevi are often similar, for genetic reasons. If one mole is slightly atypical but the patient has many others like it, that is reassuring. Beware the odd mole out, the ‘ugly duckling’. Melanomas exhibit abnormal features such as strong asymmetry, abnormal network, irregular dots and globules, blue-white featureless areas, streaks, pseudopods, eccentric black blotches, inverse pigment network, polymorphous vessels, and shiny white lines. A good starting point to see these terms illustrated is
https://www.dermnetnz.org/cme/
dermoscopy-course/pattern-analysis/
“If one mole is slightly atypical but the patient has many others like it, that is reassuring.”
Dermoscopy of seborrhoeic keratoses (SK)
These common and harmless lesions may be easy to diagnose, but their colour, shape and size vary enormously and they are often thought suspicious. Dermoscopy usually resolves uncertainty.
SKs begin as flat skin coloured or brown marks, gradually thickening over time. When thin, dermoscopy will typically reveal a pattern of curved, parallel lines which are often termed coral-like or cerebriform. As the lesion thickens, more three-dimensional structures develop such as fissures, keratin pits (comedo like openings) and keratin cysts (milia like cysts). The border may be uneven (moth eaten) but is usually sharply defined, by contrast with naevi which often fade out gently at the edge. Looped / hairpin vessels and micro papillae are also seen.
Haemangiomas are an easy win for the beginner. Dermoscopy reveals clods of red, blue or purple colour in a lace-like whitish stroma. The colour of the clods depends on the level of oxygenation of the blood in the lacunes that make up the lesion. Black indicates clotted blood.
Dermatofibromas are usually diagnosed clinically, on bi-digital palpation. However, if pigmented they can cause confusion. Dermoscopy helps by revealing a central white scar and peripheral pigmentation characterised by fine brown circles.
Basal cell cancers (BCCs) are easy to diagnose clinically when ‘classical’, i.e. an ulcerated nodule with a rolled pearly edge and telangiectatic vessels. But they are easier and cheaper to treat if picked up when smaller, and dermoscopy can help. I regularly pick up BCCs down to 3 or 4mm diameter which the patient was not aware of. For lesions on the H area of the face this may mean the difference between removal with a simple ellipse and complex surgical reconstruction, or indeed allow non-surgical treatment such as imiquimod or photodynamic therapy (PDT) which would not be possible if we wait for the lesion to declare itself at a larger size. Patients with severe sun damage and a history of multiple BCCs in my experience benefit from an annual check by a trained dermoscopist to pick up new lesions earlier. Dermoscopy has been shown to help define surgical margins of infiltrative BCCs [5].
Superficial basal cell cancers show short, sharply focused ‘serpentine’ vessels, micro erosions and sometimes angular brown structures (sometimes resembling broken spoke wheels or leaves) on a pink background. In my clinical practice, I am happy to treat with topical therapy or PDT on dermoscopic evidence, saving a biopsy.
Bowen’s disease often shows coiled, glomerulus-like vessel pattern on a red scaly plaque. Dermoscopy can usually tell Bowen’s from superficial BCC, but has little role in the diagnosis of squamous cell carcinoma (SCC), or nodular lesions in general.
Teledermoscopy
GPs often ask if they can photograph lesions and email me the images for diagnosis. Sadly, no, or at least not yet. The technology is there, and can be made to work-even for patient performed teledermoscopy [6] but organisational and political issues get in the way. Objections include confidentiality, consent and image quality. These are legitimate concerns, but can be addressed given the will. There are benefits where patients have a difficult journey to hospital, and this isn’t just prisons, highlands and islands, but frail elderly coming by ambulance transport.
Rising melanoma rates plus GPs’ limited lesion recognition skills and fear of being sued is driving up urgent skin cancer referrals, and dermatology services are feeling stressed. Many lesions seen on the urgent cancer pathway are obviously benign and could have been screened out by basic dermoscopy in primary care. It seems logical to train at least one GP per practice to use basic dermoscopy as a triage tool. We know this is achievable [4].
The future of dermoscopy?
Teledermoscopy could reduce avoidable visits to secondary care [7].
Various patient-operated Smartphone apps have been rolled out, including some designed to be used with cheap dermoscopes. Some systems send patient-taken images to a distant specialist, who may then advise a face to face consultation or surgery. Apps based on patients photographing their own naevi may be more practical, since lack of documented change over three to six months is easy enough to monitor (most melanomas change visibly over three to six months).
Artificial intelligence (AI) based devices with vast computing power are in development. I find this ironic, when medical students spend perhaps half a day total learning about skin lesions and few GPs get much more than that over a lifetime. If doctors were better trained, we wouldn’t need such gadgets.
In conclusion
The skin cancer epidemic has serious resource and training implications. A multidisciplinary response begins with universally good enough lesion recognition skills. Dermoscopy is a valuable technique for triaging skin lesions which could be rolled out as a primary care triage tool. It has skin cancer management applications such as selecting BCCs for topical therapy and defining surgical margins. Britain is behind the game globally and needs to catch up. Many free online learning resources exist, including excellent YouTube videos by board members of the International Dermoscopy Society.
References
1. Skin cancer mortality statistics. Cancer Research UK.
http://www.cancerresearchuk.org/
health-professional
/cancer-statistics/statistics-by
-cancer-type/skin-cancer/mortality
Last accessed March 2018.
2. Vestergaard ME, Macaskill P, Holt PE, Menzies SW. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol 2008;159(3):669-76.
3. 5th World Congress of Dermoscopy.
https://dermoscopy-ids.org/
5th-world-congress-of-dermoscopy/
Last accessed March 2018.
4. Argenziano G, Puig S, Zalaudek I, et al. Dermoscopy improves accuracy of Primary Care Physicians to triage lesions suggestive of skin cancer. J Clin Oncol 2006;24(12):1877-82.
5. Carducci M, Bozzetti M, De Marco G, et al. Usefulness of margin detection by digital dermoscopy in the traditional surgical excision of basal cell carcinomas of the head and neck including infiltrative/morpheaform type. J Dermatol 2012;39(4);326-30.
6. Kassianos AP, Emery JD, Murchie P, Walter FM. Smartphone applications for melanoma detection by community, patient and generalist clinician users: a review. Br J Dermatol 2015;172(6):1507-18.
7. Manahan MN, Soyer HP, Loescher LJ, et al. A pilot trial of mobile, patient-performed teledermoscopy. Br J Dermatol 2015;172(4):1072-80.
Readers wanting to learn more about dermoscopy might be interested in attending the Dermoscopy International Refresher and Update Day Conference on 28 November 2018 in Fareham, Hampshire, UK. With visiting French expert Professor Luc Thomas and UK speakers, this meeting is ideal for dermatologists, plastic surgeons, GPwSIs and all health professionals who regularly use dermoscopy for lesion recognition and skin screening. Details and booking form can be found on www.dermoscopy.wordpress.com or by emailing stevehayes272@gmail.com
Declaration of competing interest:
The author teaches lesion recognition and dermoscopy skills for reward and blogs at www.dermoscopy.wordpress.com
COMMENTS ARE WELCOME