Having full lips is commonly associated with beauty and youthfulness. This is the reason why lip enhancement is one of the most frequently requested procedures. The complete perioral frame should be taken in to consideration to achieve a more natural result.
To be more specific, the assessment for lip augmentation should be analysed with regards to the following parts: the vermillion border, the philtrum columns, the three tubercles of the upper lip and the two tubercles of the lower lip, the Cupid’s bow, the oral commissures, the nasolabial and melomental folds, the mental crease and the upper lip wrinkles.
Before focusing on these structures however, it is important to check if there is any asymmetry on the lip, the position of the teeth, maxilla and mandible. This examination should be done with the face in rest but also with dynamic movements, to see if there is any muscular hyperactivity that could be corrected with botulinum toxin.
The importance of lip ratio
To achieve a natural result, the lower lip should appear always slightly bigger than the upper one. The ideal vertical height ratio of the upper lip to the lower lip should be 1:1.6.
Aside from that, it has to be clear for the patient that lip enhancement is performed in a personalised manner. There are a variety of approaches, related to age, gender and ethnicity. In case of ageing, proportions of different parts of the lip will change. Besides the loss of volume, ageing will lead to lengthening of the cutaneous upper lip and flattening of the philtrum columns.
Figure 1: Illustration demonstrating the placement of canulas to face.
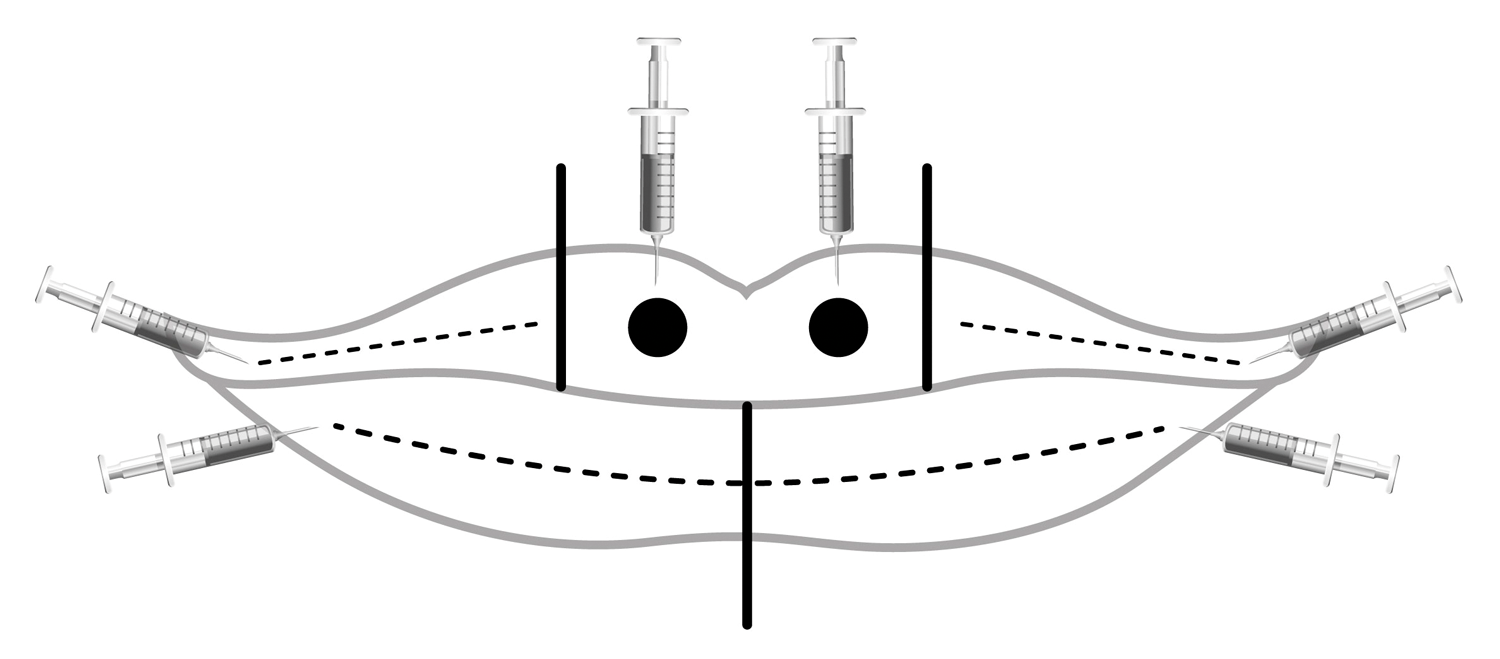
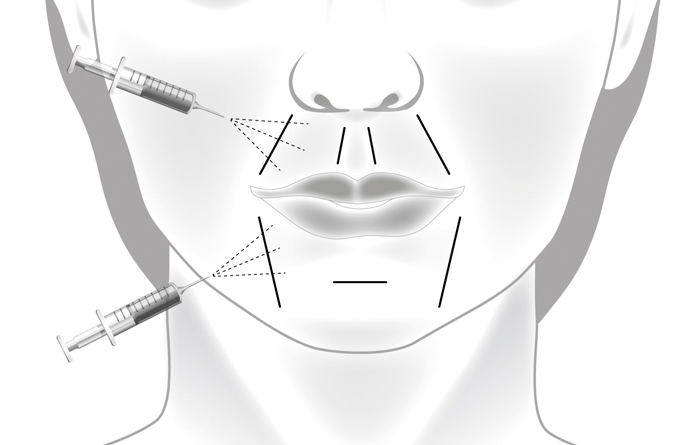
Figure 2: Illustration demonstrating the placement of needles to lips.
Lip enhancement product and techniques
From a safety point of view, I recommend hyaluronic acid (HA) as a filler. Caution must be taken to avoid intravascular injection and overcorrection.
1. Lip body and upper lip wrinkles: Aliaxin® Fine Lines & Lips (FL) is my HA filler of choice, as it has a high cohesivity, and specific rheological properties, which facilitates a better tissue integration. It is possible to inject this in a more superficial level, thereby achieving a great hydration effect and a natural lip plumping.
2. Upper lip: is divided into three parts; the lateral thirds are injected with a 30G needle that is approached laterally through the body of the lip up to the lateral tubercle. The product is then injected in a linear retrograde way using approximately 0.2ml per third of the upper lip. In the middle third two boluses of 0.05/0.1ml are injected to help to define the Cupid’s bow and pronounce the middle tubercle.
3. Lower lip: is divided with a medial line and the two parts are injected also laterally in the body of the lip until the tubercles are reached, again using linear retrograde technique for product injection of 0.25-0.3ml per half of the lower lip. In case a patient requires an above average emphasis on lip volume increase, Aliaxin® Global Performance (GP) can be used with the same technique and a 27G needle.
4. Upper lip wrinkles: I use Aliaxin® FL injected with ‘Blanching technique’. This is a technique where the needle has an angle at the insertion into the skin from about 10° to the skin surface. Blanching is a technique like serial puncture technique; make sure that the skin is held completely taut and the command of the needle is pointing upwards when inserting the needle.
Advance a single point of the needle into the skin, just below the epidermis. Introduce a small amount of the product with each injection, and give constant pressure without putting excessive force on the plunger and stop when the skin is saturated. With this technique you can see the ‘blanching effect’ (the whitening of the skin). Massage the treated area cautiously to ensure that the product spreads evenly.
5. Nasolabial and melomental folds: I use a 25G cannula, approaching with a retrograde fanning technique and a firm product, Aliaxin® GP, which can adapt to facial expressions but has more volumising capacity. Approximately 1-2ml of product is injected per session.
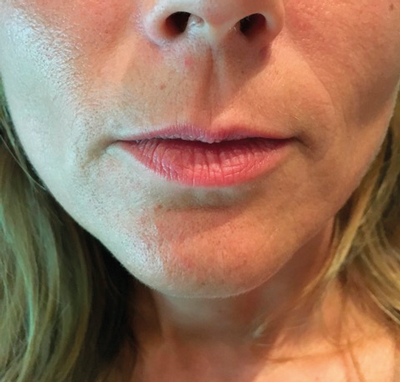
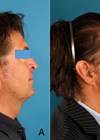
Figure 3: Patient before treatment.
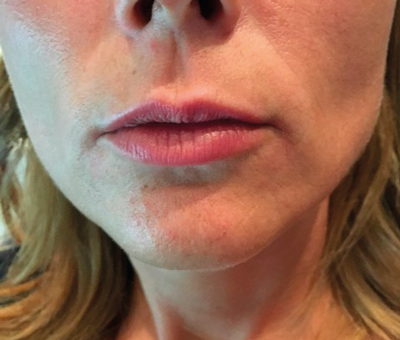
Figure 4: Patient after treatment.
Declaration of competing interests: None declared.
COMMENTS ARE WELCOME