GLP-1 receptor agonists (GLP-1) and combined GLP-1/gastric inhibitory polypeptide (GLP-1/GIP) agonists have emerged in recent years to promote weight loss [1]. The incretin mechanisms of GLP-1 agonists help to regulate blood glucose levels and facilitate satiety, which contributes towards weight loss [2].
These prescribed medications encourage the production of insulin, inhibit glucagon production from pancreatic α-cells, and reduce pancreatic β-cell apoptosis [3]. This class of drug subsequently suppresses appetite and is accompanied by delayed gastric emptying and has Food & Drug Administration (FDA) approvals for type 2 diabetes and obesity [4].
An increasing number of observed and often rapid skin changes are frequently reported, which include dehydration and sagging, as well as thinning of skin, which is often most notable on the face [5]. It is postulated that GLP-1 receptor agonists may affect other types of skin cells, which may contribute to the accelerated features of facial ageing [6]. In addition, other proposed GLP-1 mechanisms include targeting adipose-derived stem-cells (ADSC) and fibroblasts where these receptors are present on the cell surface [6]. This stimulation is thought to trigger a destructive cascade, which leads to the production of reactive oxygen species leading to direct oxidative damage on fibroblasts. Furthermore, the effects of GLP-1 on ADSCs is thought to negatively impact the production of oestrogens from dermal white adipose tissues, which further inhibits the regenerative capacity of fibroblasts [7].
This case study will outline the results of a topical cosmetic product formulated with specific ingredients to address a number of dermatological concerns which patients experience during GLP-1 therapy. Vol.U.Lift (Image Skincare) combines a unique blend of ingredients, including L-Ornithine amino acid, which has shown benefits in increasing fatty tissue density through three key mechanisms:
- The activation of adipogenesis (transformation of pre-adipocytes into adipocytes).
- The activation of lipogenesis (fatty acids synthesis).
- The inhibition of lipolysis (breakdown of lipids into free fatty acids) [8].
In addition, the formulation contains bakuchiol which is a popular alternative to retinol, and well tolerated with comparable benefits regarding collagen synthesis (I, III, IV & VII) [9]. The topical is formulated with HA silanol to support hydration levels in the skin, as well as plant collagen-fragment, and angiozathos flavidus plant extract (kangaroo paw extract), which is a botanical rich in flavonoids, tannins and phenolic acids, procollagen 1 production and tenascin-X synthesis, which is key for supporting skin integrity. The technology includes a patent pending blend (XOSMTM), which is designed to amplify the antioxidant effects of ectoin, vitamin C and astaxanthin-rich micro-algae [4].
Case report
A 79-year-old caucasian female presented to clinic. She had no significant medical history, but had recently been prescribed tirzepatide (Mounjaro®) once weekly injections (2.5mg dose) for weight management and had been receiving therapy for two months. During the consultation it was noted that she had: no known allergies; was a non-smoker; had no prior, or existing, skincare regime; and no prior medical aesthetic or skin rejuvenation procedures. Her skin ageing (face and neck) was assessed as Glogau grade III.
Treatment aim/patient concerns
The patient’s main concern was the rapid onset of skin laxity and thinning around her neck area, which she had developed since commencing tirzepatide therapy. In addition to the tissue thinning there was evidence of textural roughness and deydration, as well as mild redness, inflammation and photodamage. In light of the fragility of the skin, and following a detailed consultation, and discussion around suitable treatment options, the recommendation was to commence a daily topical treatment (Vol.U.Lift). The treatment aims were to strengthen and improve the skin’s resilience, by boosting collagen, hydration and overall firmness, as well as nourishing the skin barrier. In the author’s experience, commencing a patient on this type of topical is a highly effective and simple method to prime and prepare the skin ahead of more invasive medical aesthetic procedures, but may also be used in conjunction with a variety of treatments. The patient underwent once daily use of Vol.U.Lift for a total of four weeks (baseline and post-treatment images).
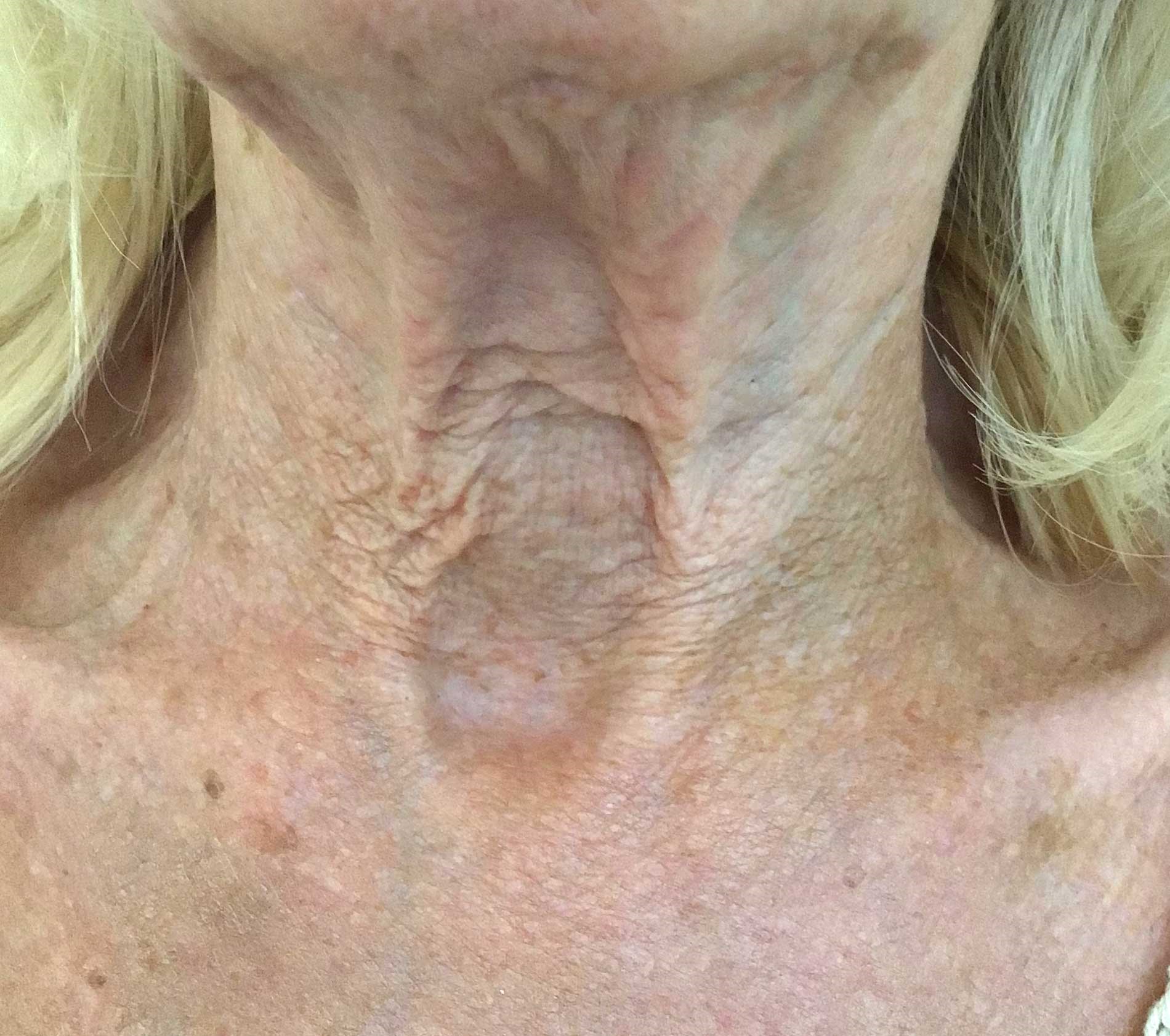
Pre treatment.
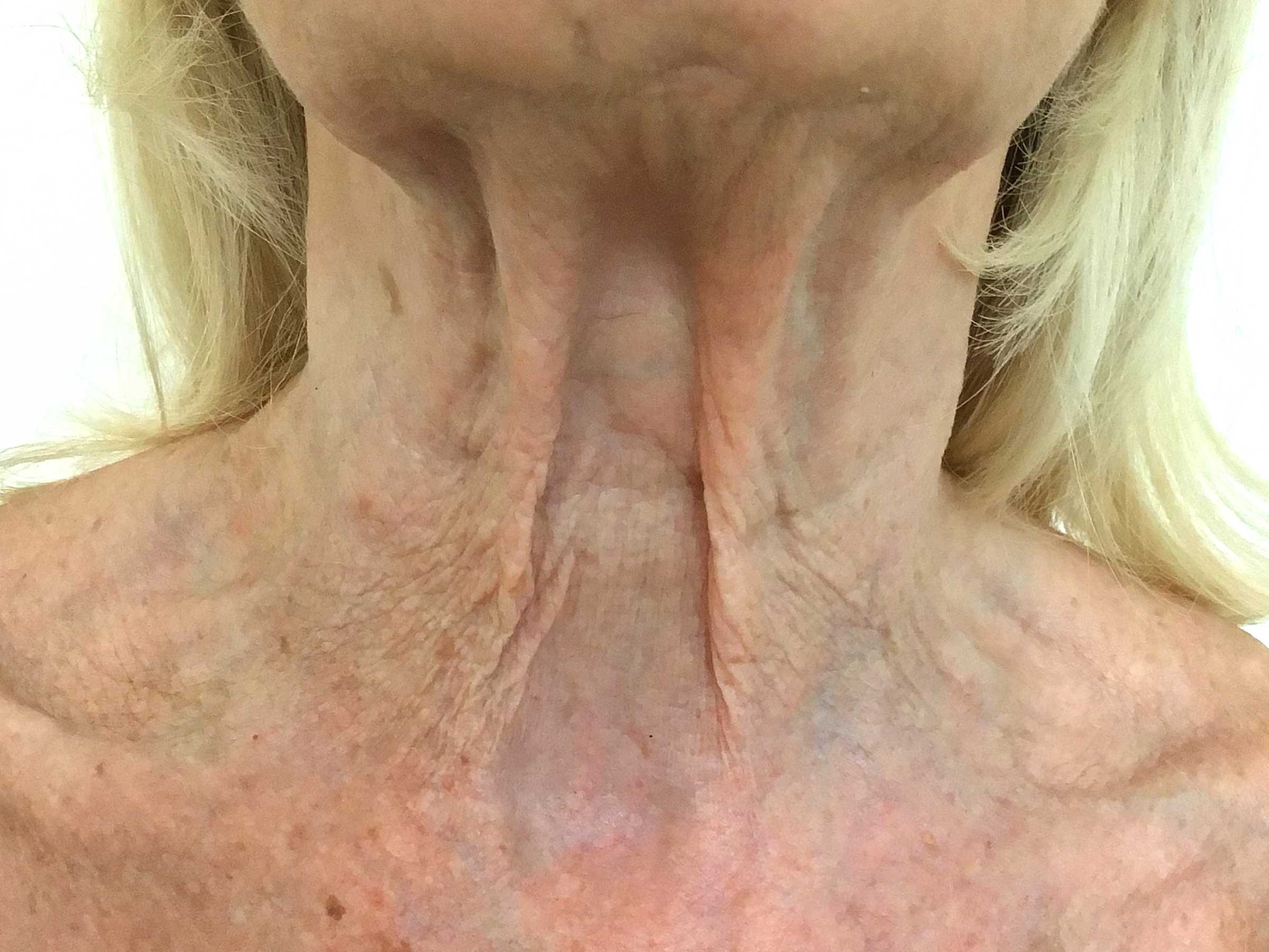
Post treatment.
Results
Self-reported outcomes exceeded anticipated results. The patient commented how rapidly she began to feel improved firmness and hydration in her skin, and boosted confidence in choice of clothing. There was a visible improvement across a range of existing concerns, including improved elasticity and firmness, reduced redness as well as photodamage. No adverse events were reported and the patient is keen to continue to hopefully anticipate long-term cummulative benefits.
Conclusion
The rise of GLP-1 use is anticipated to continue, and medical aesthetic practitioners should remain mindful of an expanding body of literature exploring the mechanisms which appear to underpin these unique and rapid tissue changes. These prescription therapies are an emerging and niche area of medicine, but it is likely that the number of patients on GLP-1 treatment experiencing accelerated skin changes will increase. Arguably a topical formulation which is designed to address such changes could be a key component of the patient journey-either as a standalone topical, or to amplify a combination approach.
References
1. Moore PW, Malone K, VanValkenburg D, et al. GLP-1 agonists for weight loss: pharmacology and clinical implications. Adv Ther 2023;40(3):723–42.
2. Haykal D, Hersant B, Cartier H, Meningaud JP. The role of GLP-1 agonists in aesthetic medicine: exploring the impact of semaglutide on body contouring and skin health. J Cosmet Dermatol 2025;24(2):e16716.
3. Persson C, Eaton A, Mayrovitz HN. A closer look at the dermatological profile of GLP-1 agonists. Diseases 2025;13(5):127.
4. Nguyen N, Aguilar A, Afzal N, et al. Topical volumizing cream improves facial volume and skin health in adults with rapid weight loss from pharmacologic (GLP-1/GIP agonists), surgical, or behavioral interventions. J Cosmet Dermatol 2026;25(1):e70681.
5. Burke OM, Sa B, Cespedes DA, Tosti A. Dermatologic implications of glucagon-like peptide-1 receptor agonist medications. Skin Appendage Disord 2025;11(5):416–23.
6. Paschou IA, Sali E, Paschou SA, et al. GLP-1RA and the possible skin aging. Endocrine 2025;89:680–5.
7. Patino W. Thomas A, Jain S, et al. A review of glucagon-like peptide-1 in dermatology. J Clin Aesthet Dermatol 2025;18(3):42–50.
8. Ito N, Seki S, Ueda F. Effects of composite supplement containing collagen peptide and ornithine on skin conditions and plasma IGF-1 levels: a randomized, double-blind, placebo-controlled trial. Mar Drugs 2018;16(12):482.
9. Bluemke A, Ring AP, Immeyer J, et al. Multidirectional activity of bakuchiol against cellular mechanisms of facial ageing – experimental evidence for a holistic treatment approach. Int J Cosmet Sci 2022;44(3):377–93.
Declaration of competing interests: None declared.





