With the ever increasing demand for injectable treatments, it is important to keep abreast of new developments in the field. International expert Michael Gold reviews the new toxins due to be hitting the market in the next year.
We are very fortunate in cosmetic and aesthetic dermatology that we have an array of minimally invasive cosmetic treatments that have grown in prominence and numbers over the past few years. According to the American Society of Dermatologic Surgery Survey on Dermatologic Procedures performed in 2017 [1], dermatologists performed nearly 12 million procedures in the United States in 2017, up from 7.8 million procedures in 2012. The survey found that nearly 3.7 million procedures were done with injectables consisting of neurotoxins and soft tissue filling agents, with an increase of some 79% over the past six years. Most of the neurotoxin injections occurred in patients between the ages of 40-59 years of age, 88% of the patients were female, and 12% were male, this latter marked a 9% growth rate for males receiving toxins over the past six years. Millennials, who have recently emerged as an important new market segment for cosmetic procedures, represented a doubling of the number of injectable treatments over the time period.
“Having a longer duration of effect means that our patients will be happier coming to see us less – but if the cost is more, will that have a positive or negative affect on physicians willing to purchase the new product?”
In the US there are three toxins which are used on a regular basis in the US, with Food & Drug Administration (FDA) approval. These are: BOTOX® Cosmetic (Onabotulinumtoxin A), Dysport® (Azzalure® in Europe) (Abobotulinumtoxin A), and Xeomin® (Bocouture® in Europe) (Incobotulinumtoxin A). In the US Botox Cosmetic has FDA approvals for the glabellar complex, forehead lines and the crow’s feet region. In Europe, Bocouture had the first approval for the glabellar, forehead and crow’s feet region. Dysport has approval for the glabellar and crow’s feet lines.
Botox Cosmetic had its original FDA approval in April 2002 and is marketed by Allergan. It is the market leader both in the US and internationally. The second US toxin is Dysport, which received its FDA approval in May 2009 and in the US is marketed by Galderma. It was originally developed by Ipsen Pharma, had the US rights sold to Medicis Pharmaceutical Company, which then was purchased by Valeant Pharmaceutical Company, and then sold to Galderma. The third US toxin is Xeomin, which received its FDA approval in July 2011; it is marketed globally by Merz.
These three toxins have had the advantage of being present in the US market for many years now. As noted, onabotulinumtoxinA is the market leader followed by abobotulinumtoxinA and then incobotulinumtoxinA in terms of vials used per year in the US. It is not the purpose of this manuscript to defend one product over the other; it is up to the individual clinician to determine which one of these toxins best fits their clinical experience and their practice of aesthetic medicine. What is interesting to watch, and this is truly my own experience, is to witness how some clinicians note that one toxin is superior to the other and that how their toxin of choice works faster and lasts longer than the others. I feel that this is an individual choice and that with experience, one will find the best way to utilise these toxins to make them work best for your patients.
So with three available toxins in the US market, the logical question thus arises, do we need any more toxins for us to choose from for our patients? Again, this is a question which is beyond the scope of this manuscript but a question nonetheless to consider as we are poised, at least in the US, to have at least three more toxins coming to the market over the next several years. What advantages might they have, if any? This question needs to be answered and for us, as clinicians, we need to factor into how and why we use a particular toxin. One thing that the current toxins have in their favour is the loyalty programmes that have been put into place by each of the current companies in this area. They provide discounted products for clinicians based on the amount of product purchased, as well as factoring in other products that these same companies sell to us for other cosmetic uses. These loyalty programmes are powerful and will continue to influence providers as new products come to the market place in the next few years.
The US market may see three new toxins coming to market in the next few years. Several of these toxins are already available to many that reside outside the US and are making their way through the FDA approval process now. We will now focus on these toxins, and we will share some of the information that is in the public domain for these toxins, and will not share any information that is not readily available at this time.
The first of these products primed for US approval is a South Korean toxin from Daewoong, which will, upon its approval, be known commercially as Jeuveau™ (Parabotulinumtoxin A) in the US and Canadian market; the US based company that will be bringing this toxin to market is called Evolus. The Canadian authorities granted approval of the toxin in August 2018. Currently, the Daewoong toxin is known in most parts of the world as Nabota® and in its original non-inferiority trial comparing it to Botox Cosmetic, the primary endpoint, which was maximum frown response of the toxin in the glabella area at week four, the Korean toxin showed non-inferiority as compared to Botox Cosmetic. They were also able to demonstrate non-inferiority at all other time periods of the trial, up to 16 weeks after injection of the toxins. Two US pivotal Phase III clinical studies showed that with a two-point improvement needed to show effect, compared to placebo, all of the study endpoints were met. Safety was also assessed during the studies and showed that the product was safe in all of the clinical trials that were performed. This toxin may be cleared in early 2019 for use in the US [2].
The second toxin which may receive FDA approval in the US in 2020 is made by Revance Therapeutics, Inc. and will be known as DaxibotulinumtoxinA injection. Originally, DaxibotulinumtoxinA topical gel (RT001) was being evaluated for topical use for the treatment of lateral canthal lines. The Phase II US clinical trials showed safety and efficacy to warrant Phase III studies. Unfortunately, the Phase III trial did not achieve its primary endpoints, and Revance decided not to further pursue development of RT001 topical for crow’s feet.
DaxibotulinumtoxinA for Injection (RT002) is a unique molecule that has shown safety and efficacy through the FDA’s clinical trial programme. In addition to the botulinum toxin part of the molecule, the Revance injectable toxin has a patented stabilising excipient protein in its formulation that has been shown to have some interesting properties that has allowed Revance to study daxibotulinumtoxinA for an extended duration of use as compared to the toxins that are already on or coming to the market. The Phase II clinical trials were performed in Canada and the two identical Phase III pivotal trials were performed with US and Canadian investigators looking at a ‘higher’ dose of toxin and a longer clinical endpoint than that which we have with the current toxins in the US market. In these two pivotal trials, safety and efficacy data which are available in the public domain show that daxibotulinumtoxinA, at a dose of 40 units injected into the glabellar complex, is safe and lasts upwards of six months in a significant number of patients as compared to placebo. An open labelled long-term safety study, in which patients were eligible to receive up to three treatments and were followed up to 84 weeks, confirmed the safety and efficacy data seen in the pivotal trials. Revance also recently announced that it will be initiating studies in 2019 to evaluate the use of RT002 in forehead and lateral canthal lines. Currently, Revance plans to submit a biologics license application (BLA) in the first half of 2019 with an anticipated approval in 2020 [3].
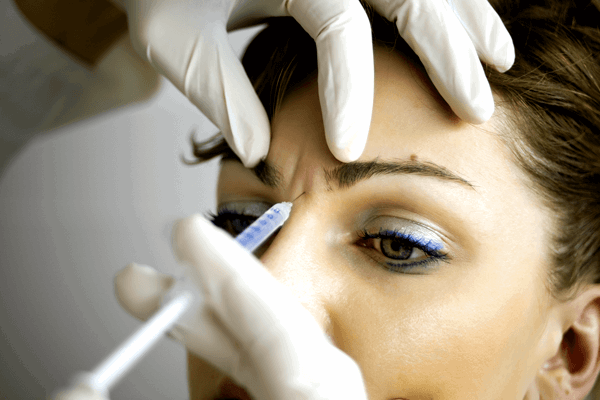
Figure 1: Treatment of the glabella area.
The third toxin currently being evaluated in the US is known as the Croma-Pharma toxin. It is also produced in South Korea by a company known as Hugel. This is known as Botulax®, amongst other names, in various parts of the world. Croma-Pharma, an Austrian-based aesthetic company, is spear-heading the US regulatory work for this toxin. The Phase II US studies showed the safety and efficacy of the product and the Phase III clinical work is underway now. No other pertinent information has been placed in the public domain for this product at this time [4].
“Do these new toxins help or hinder us as providers who regularly inject these medicines?”
Do these new toxins help or hinder us as providers who regularly inject these medicines? That is for time to tell. Perhaps a new toxin which acts similar to the toxins we have will have a price point which is lower that may attract providers to use them. Or we may find that in the clinical world, one of these works better for our patients – time will tell. But what about toxins with a longer duration than what is commercially available? This is an intriguing thought – and one which can be argued both ways. Having a longer duration of effect means that our patients will be happier coming to see us less – but if the cost is more, will that have a positive or negative affect on physicians willing to purchase the new product? Allergan, Galderma, and Merz are all looking, or will be looking, at ‘stronger’ doses of toxin for a longer duration of effect; so perhaps, the Revance toxin is on to something exciting for our patients. Again, we will answer this question over the next few years, but it is intriguing, as are the new toxins that will be coming to the US market soon.
References
1. https://www.asds.net/
portals/0/PDF/procedure-survey
-results-presentation-2017.pdf.
2. https://investors.evolus.com/
news-releases/news-release-details/
evolus-receives-conditional-fda
-acceptance-jeuveautm-brand-name
3. http://www.revance.com/pdfs/
Sakura_1_2_TLR_Presentation_at
_IMCAS_3_Feb_18_Gary_Monheit.pdf
4. https://at.croma.at/
home-en-ca/news-and-events/news/
(All links last accessed December 2018.)
Declaration of competing interests: Dr Gold has performed research for Revance and Croma-Pharma.
COMMENTS ARE WELCOME




