This is the first part of a two-part article looking at the challenges of reconstruction of complex defects in the torso. The articles are based on a presentation given at the World Union of Wound Healing Societies held in Yokohama in 2012. The presentation has been updated and refined to reflect evolving ideas in this challenging field of reconstruction.
A good teaching mantra is the following: a wound is a discontinuity in tissue integrity; healing is the process of restoring that integrity; healing (in the skin) can occur by two biological processes, regeneration and repair; regeneration results in no scarring, repair results in scarring. This sets the scene for talking about surgical reconstruction which can involve both biological processes. The emphasis in these articles is complex wounds and the term complex needs some definition.
Wounds can be complex because of:
- Size
- Site
- Structural defects
- Specific patient factors.
Reconstructive surgeons are typically introduced to the concept of the ‘reconstructive ladder’ (Figure 1). The idea being that the more problematic the wound the higher up the ladder the surgeon has to climb [1].

Figure 1.
The term reconstructive ladder was coined in the 1980s by those two giants of plastic surgery Stephen Mathes and Foad Nahai. A whole generation of plastic surgeons have grown up paying lip service to the concept but not really knowing what it means. It is really a conceptual tool to help organise thought rather than influence practice. A ladder without support is inherently unstable unless you place it on the ground and then we see a different concept, a spectrum (Figure 2).
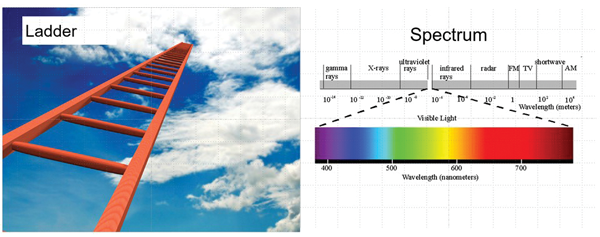
Figure 2.
I am sure that Stephen Mathes, who was a great thinker in plastic surgery, would agree with this change of perspective. We can now look at the range of options available to the individual surgeon and put these in the context of experience, resources and skills. As an example, there is no doubt that a simple microsurgical free tissue transfer is technically a much simpler procedure than a large full thickness graft. But more of that later. What about the specific anatomical challenges of the trunk and the pelvis that will influence the reconstruction?
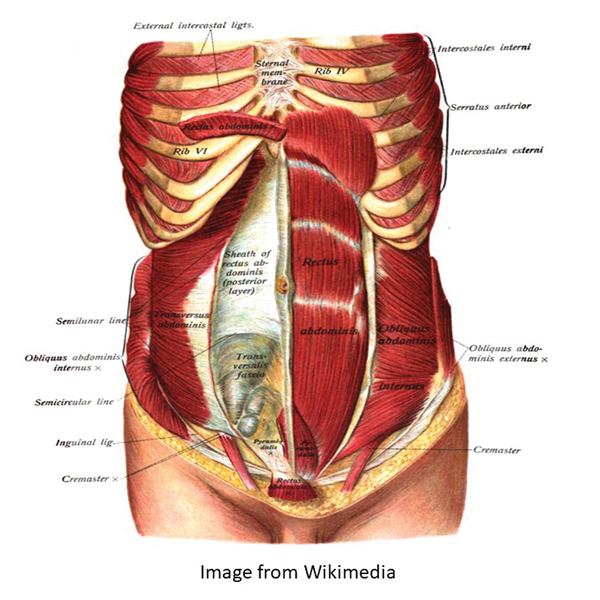
Figure 3.
In the trunk, consideration has to be given to the thoracic and abdominal viscera with functional compartmentalisation (Figure 3), whilst in the pelvis, consideration has to be given to the passage of urine and faeces (Figure 4). Diversion may be necessary for either, or both, and this may be temporary or permanent.
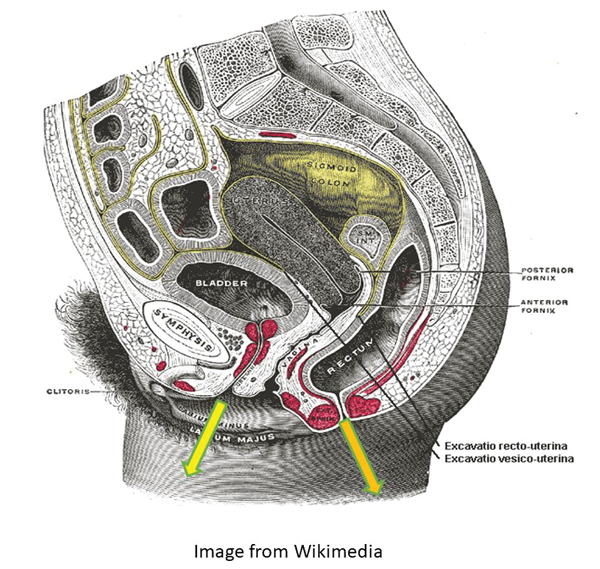
Figure 4.
As with all reconstructions the key to success is encapsulated in the famous words of Robert Acland, “Preparation, preparation and preparation”. In this context, there are four important goals:
- Optimise the patient
- Optimise the wound
- Plan the surgery
- Prepare the team.
With this background we can start to consider the reconstructive spectrum and begin with cells.
Cells
When considering wounds in the skin, burns surgeons recognise the best dressing is skin itself. The most important part of the skin for this purpose is the outer layer, the epidermis and within the epidermis the most important layer is the stratum corneum. This is the outer layer of dead cells which has a primary role as a protective barrier. Before considering skin grafts we want to look at the role of cells which are cultured in the laboratory and can be applied either as a confluent sheet or as a suspension of cells.
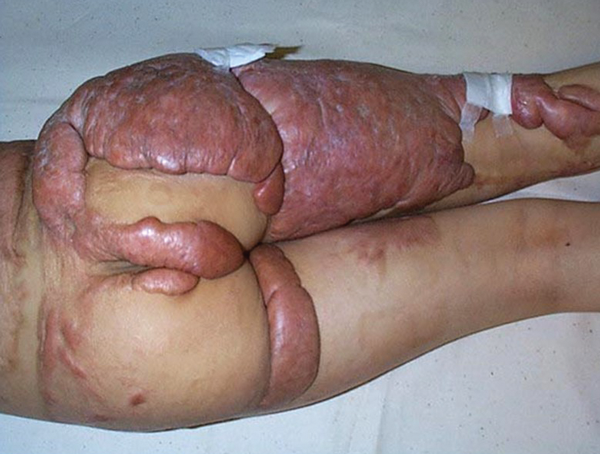
Figure 5.
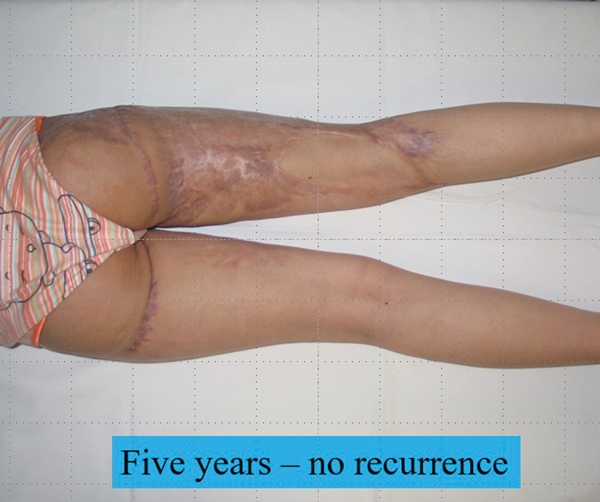
There is no doubt that the extreme antigenicity of the epidermis means that autologous cultures are required if the cover is going to be permanent. There is, however, considerable work underway to look at the potential for allogenic sources, particularly of foetal origin to act as universal donor cells for temporary cover. There is a safe, abundant, and an ethically acceptable source of such cells, the human umbilical cord lining cells. It is evident though that using cells to resurface a wound can have a more profound biological effect on tissue response and this is illustrated in the case of a young girl with an extensive post burn keloid. Why do we call this a keloid? It is raised with rolled edges and no associated contraction (Figure 5).
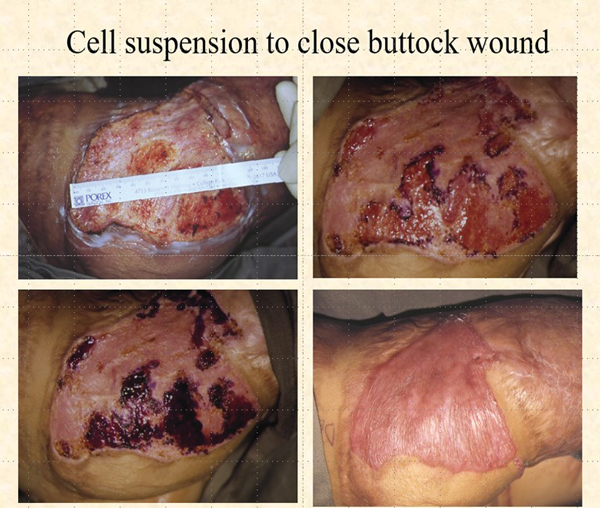
Figure 6.
This remarkable case was reported some years ago in Plastic and Reconstructive Surgery [2]. A wound was surgically created on the right buttock by excising the keloid to the level of the surrounding skin. This dermal / scar network was then resurfaced with a suspension of autologous keratinocytes cultured from a biopsy of normal skin. It took four weeks for the wound to be completely resurfaced (Figure 6) and then the most remarkable thing happened. The keloid did not recur and the scar in the non-operated parts began to resolve. Five year follow-up showed no evidence of keloid and whilst scarring remained there was no restriction of growth and no contraction (Figure 7).
Figure 7.
Split thickness grafts
The use of split thickness grafts is one of the most common procedures in reconstructive plastic surgery, particularly in acute burns care. Burn wounds become more complex when the burn is full thickness and a fascial excision is required. The following series of slides shows the excisional technique of using a burn to treat a burn. Caution must be expressed that in the more regulated healthcare practice today the use of a DIY suction diathermy would most probably not be allowed but the following two slides show such a device in preparation and in action (Figures 8-9).
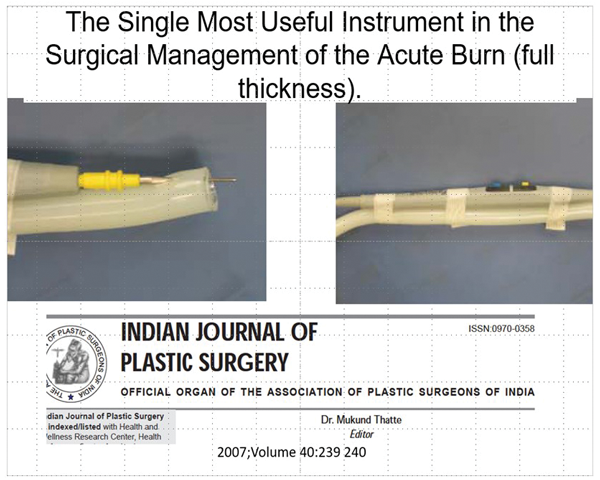
Figure 8.
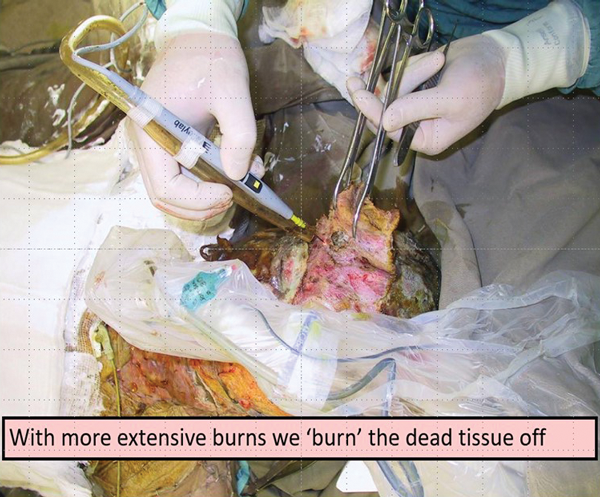
Figure 9.
The following two slides show a before and after of such a fascial excision. In the acute phase, this early aggressive total burn wound excision does lead to a cessation of the systemic inflammatory response with a dramatic decrease in inotrope load (Figures 10-11).
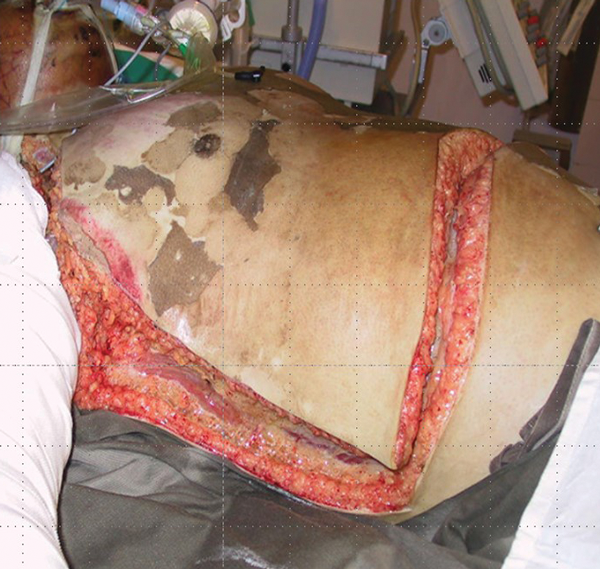
Figure 10.
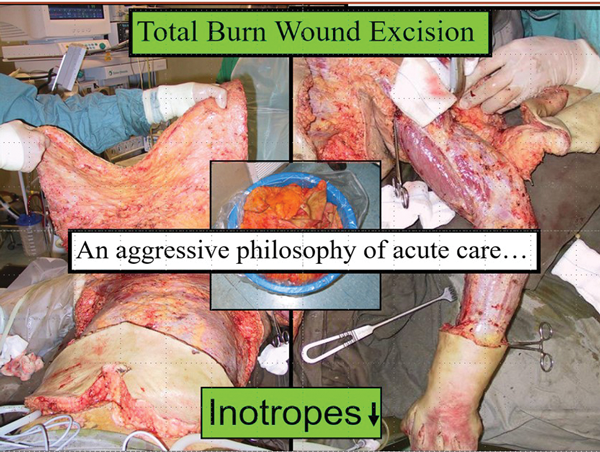
Figure 11.
Of note, look at how the transverse incision is made to decompress the chest wall and disconnect the thorax from the abdomen. This must be below the costal margin and not as has been handed down from textbook to textbook, above the costal margin [3].
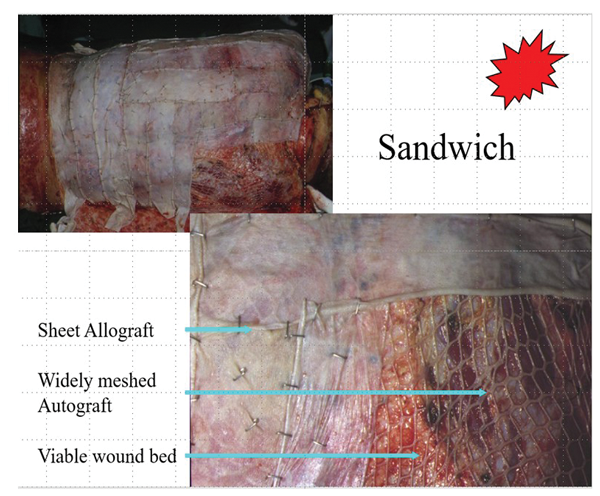
A complex wound has been created and how can this be closed? A split skin graft. Of course, how this is used will depend on the available donor sites. When a patient has extensive burns there will be limited donor skin. It is beyond the scope of this article to talk about the management of the 60+% body surface area (BSA) burns but in the 40-60% BSA range, skin can be harvested and then widely meshed. If applied directly the tissues exposed by the interstices of the mesh would desiccate. This can be prevented by overlaying sheet cadaver allograft. This technique is called sandwich grafting and has stood the test of time. The slides show the sandwich and also the before and after of a posterior trunk burn treated with this technique (Figures 12-13).
Figure 12.
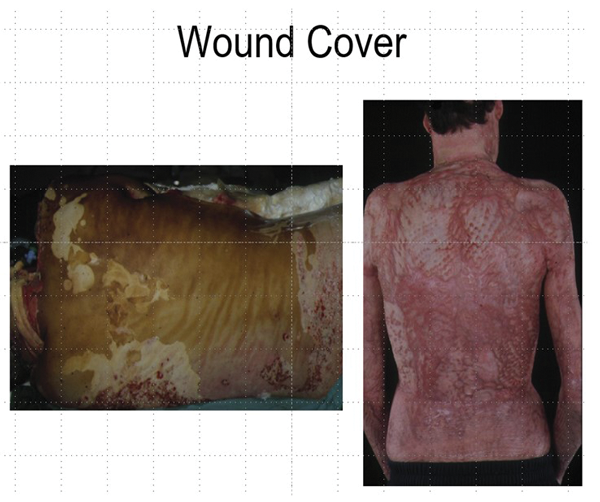
Figure 13.
Full thickness grafts and tissue engineering
It will become apparent to the reader that this article is not following the traditional reconstructive ladder but is intermingling techniques, which is more reflective of real-life practice. The challenge in this next illustrative case is less the full thickness graft and more the management of the donor site from which it is harvested. The patient illustrated is a young boy who developed post burn scarring of the face and neck. This exhibited features of both keloid and hypertrophic scarring, which is a combination not uncommonly seen in children but less so in adults. This is probably a reflection of the biodiversity between adults and children, i.e. they are different in response to similar stimuli. The slides show the preoperative view of the neck, the large full thickness graft applied and the early postoperative view (Figures 14-16).
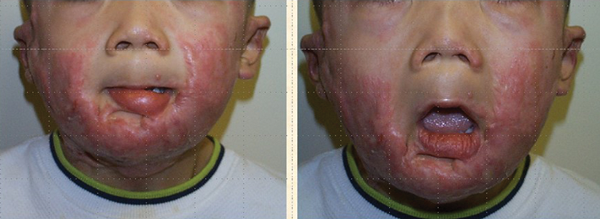
Figure 14.
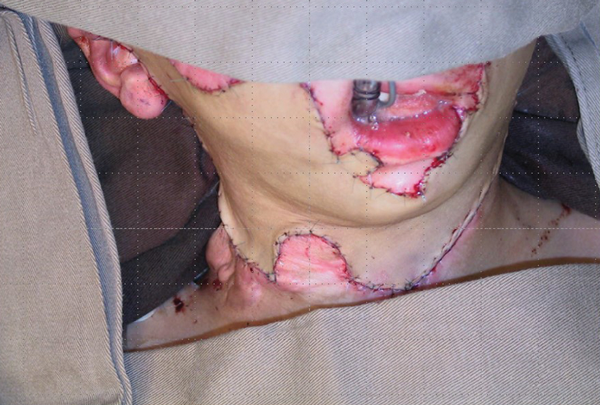
Figure 15.
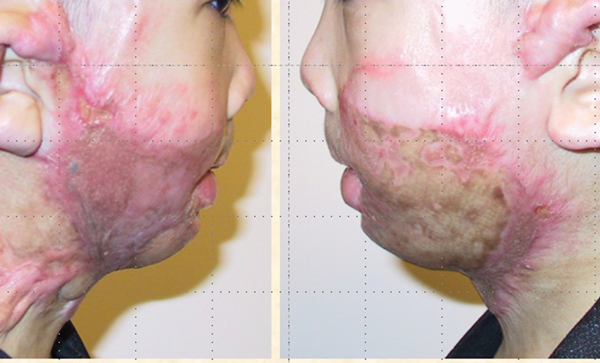
Figure 16.
The focus is not on the neck but the abdominal donor site, which is far too large to close directly (Figure 17).
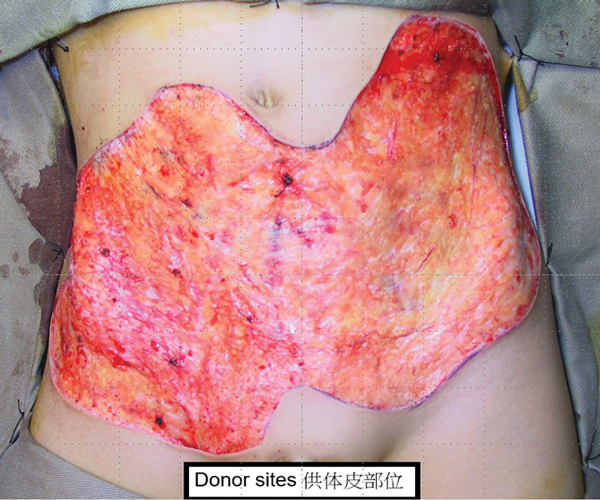
Figure 17.
“When looking at the various products available it is important to understand how they work and how they behave over a period of time”
How would we approach this if we were going to use the traditional reconstructive ladder? We could use a meshed split thickness graft as described above, but we have more strategies to draw upon and in this case, tissue engineering. When looking at the various products available it is important to understand how they work and how they behave over a period of time. The first commercial product was marketed as ‘Integra dermal regeneration template’. Again, it is beyond the scope of this article to go into the scientific background of the development of this product but essentially it is a bilaminar structure with a protective silicon layer overlying a highly organised matrix of Type I bovine collagen woven in a bed of chondroitin sulphate. It is relatively inert biologically so does not stimulate a typical inflammatory response. Instead macrophages are attracted into the bovine collagenous matrix and as they eat their way through it they are followed by fibroblasts which lay down an auto collagenous matrix not dissimilar in morphology to a normal dermal matrix. This then becomes vascularised and in a second stage the silicon is removed and replaced by an ultrathin split thickness graft.
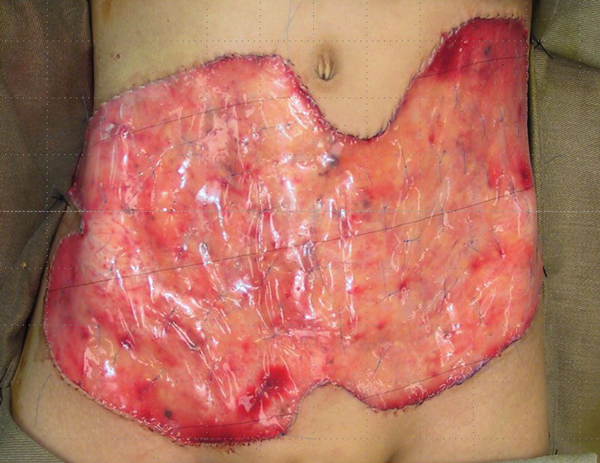
Figure 18.
Figure 18 shows the Integra in place with running sutures around the edge and multiple quilting sutures to prevent haematoma collection and shearing. As experience grew with the use of Integra for defects on different parts of the body it became apparent that the new auto collagenous dermal like matrix was not stable. When placed on an extensor surface it would stretch, when placed on a flexor surface it would contract. It takes some six to nine months for the matrix to stabilise. It was this property of malleability that was being exploited. Using serial photographs and computer planimetry a progressive reduction in the size of the donor scar could be followed. Again, with time and patience the scar can reduce to the size where excision and direct closure can be achieved (Figure 19). It must be emphasised that this was using a tissue engineered dermal regeneration template to close an anterior abdominal wall defect which is essentially a flexor surface. Posterior defects are different and this is illustrated in this case of a giant hairy nevus which has been excised and the defect is closed with Integra (Figures 20-21).
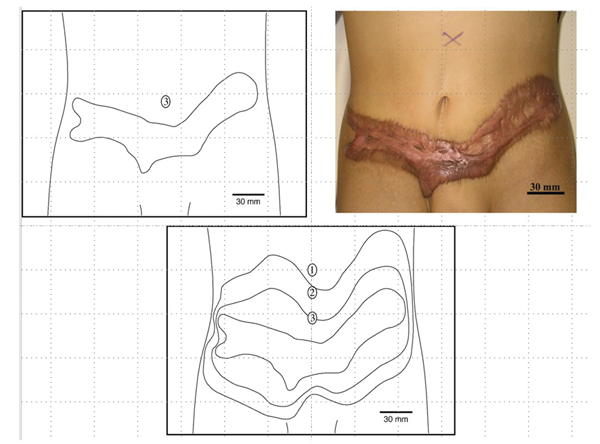
Figure 19.
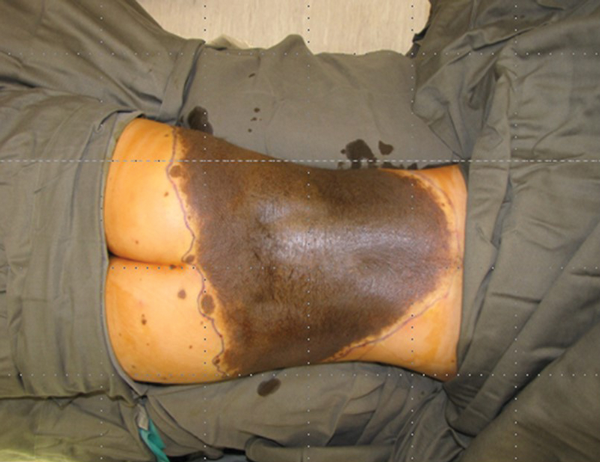
Figure 20.
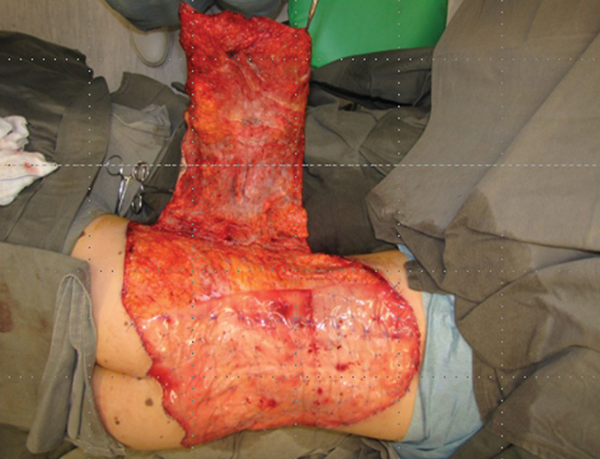
Figure 21.
The sequence of slides show the Integra in place and then after two to three weeks the colour changes to an almond yellow. The silicon layer is removed and a graft is placed that completes the reconstruction of the full thickness skin defect with a bilaminar new skin (Figures 22-25). There will be no contraction of the reconstructed area which will undergo a progress reduction in colour to eventually become similar to normal skin. Of note, this reconstructed area will grow with the child and the only difference from normal skin would be the lack of appendageal structures.
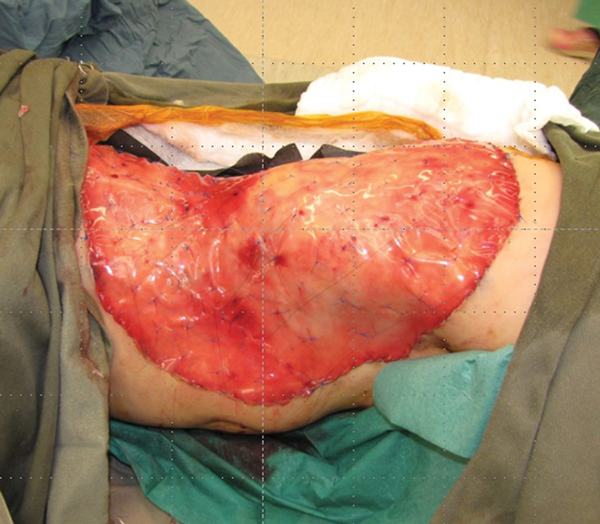
Figure 22.
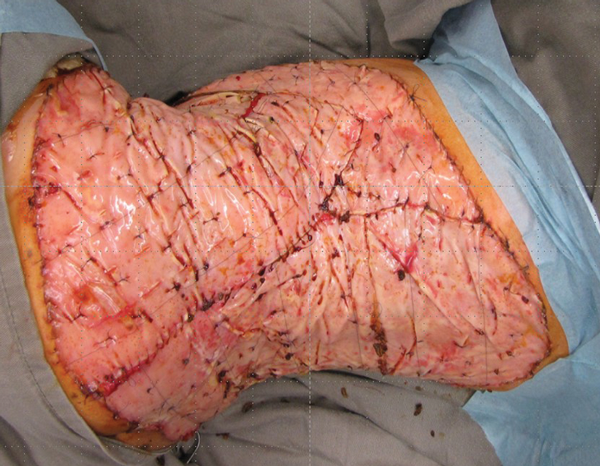
Figure 23.
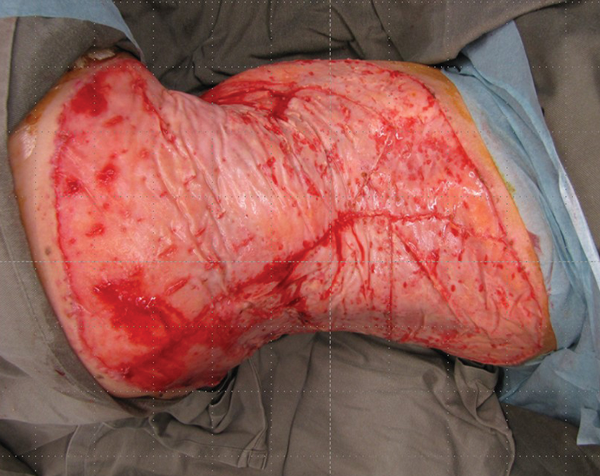
Figure 24.
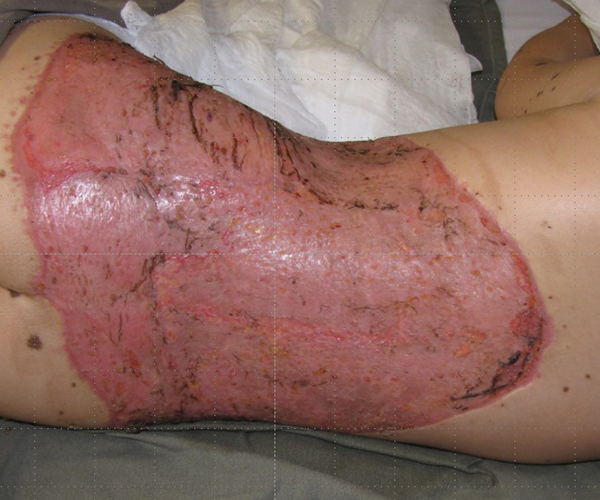
Figure 25.
Tissue expansion
Moving on from tissue engineering we come to tissue expansion. It is of interest that this does appear in the traditional reconstructive ladder between flaps and grafts. Tissue expansion of the abdominal wall during pregnancy is as old as life itself and yet this ability to create new tissue only became part of the surgical armamentarium in the 1980s. There are two main strategies that exploit the ability to expand tissue, one is to make space to fill and the other is to make new tissue to fill defects. Thus one of the early uses of tissue expansion was in breast reconstruction after mastectomy. This is now part of the established spectrum of options for routine clinical application. The other application is where a defect is going to be created and new skin needs to be produced to close the defect. There was quite a debate in the early days of tissue expansion regarding whether the new skin was being created or the old skin was being stretched.
Time is a very important factor. When skin is rapidly expanded it does remain elastic in nature and can retract. This lesson was learnt in breast reconstruction and so the policy of over expanding and allowing the skin to stabilise before removing the expander and replacing it with a permanent prosthesis became standard procedure. In the example shown of the preoperative plan two expanders of different size and configuration will be used to close the defect after the excision of this giant naevus in an adult patient (Figure 26).
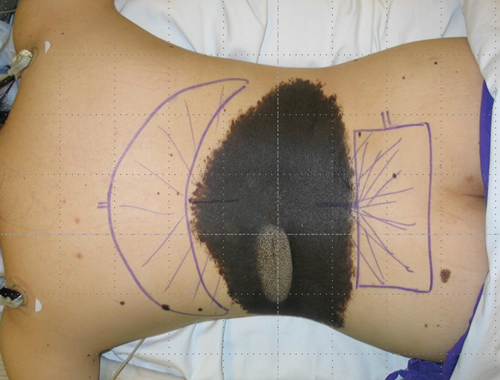
Figure 26.
This concludes the first part of this article and reflects a rather different spectrum from the traditional ladder. Starting with cells, we have moved onto grafts, then tissue engineering and have arrived at tissue expansion. In all cases we are looking not only at the intervention itself but also the impact of time on the outcome.
References
1. Boyce DE, Shokrallahi K. ABC of Wound Healing. BMJ 2006;332:710-2.
2. Burd A, Chan E. Keratinocyte-keloid interaction. Plast Reconstruct Surg 2002;110(1):197-202.
3. Burd A, Noronha FV, Ahmed K, et al. Decompression not escharotomy in acute burns. Burns 2006;32(3):284-92.
Declaration of competing interests: None declared.
See here for Part 2 of this topic.
COMMENTS ARE WELCOME