The key to maintaining a youthful appearance is often found in restoring a well-defined periorbital area, characterised by healthy tissue volume (fullness) in the infraorbital region and a clear eyelid crease devoid of excess skin. The use of hyaluronic acid (HA) dermal filler to rejuvenate the infraorbital area has increased in recent years. Formulations and techniques have been refined, giving patients a safe and effective alternative to surgical blepharoplasty. HA is a versatile injectable filler which can be modulated to alter its hardness, longevity and lifting capability.
HA dermal fillers are ideal for use in the infraorbital area as this naturally occurring glycosaminoglycan disaccharide is hydroscopic, restoring pliable volume, while posing little risk of immunologic response and rejection as it is biocompatible across species [1].
HA has long-lasting effects as it is slowly degraded by the enzyme hyaluronidase over a series of months [2]. A further advantage of HA is that complications such as product migration and over-correction can be addressed by enzymatic degradation of the dermal filler [3].
Anatomy and pathogenesis of ageing
Safe and effective rejuvenation of the infraorbital region relies on a thorough understanding of the anatomy of this area, the foundation of which is the bony orbit. Over the bone, the infraorbital region (Figure 1) consists of [4]:
- The skin
- Superficial fat compartment
- Orbicularis oculi muscle (OOM)
- Suborbicularis oculi fat (SOOF)
- Periosteum
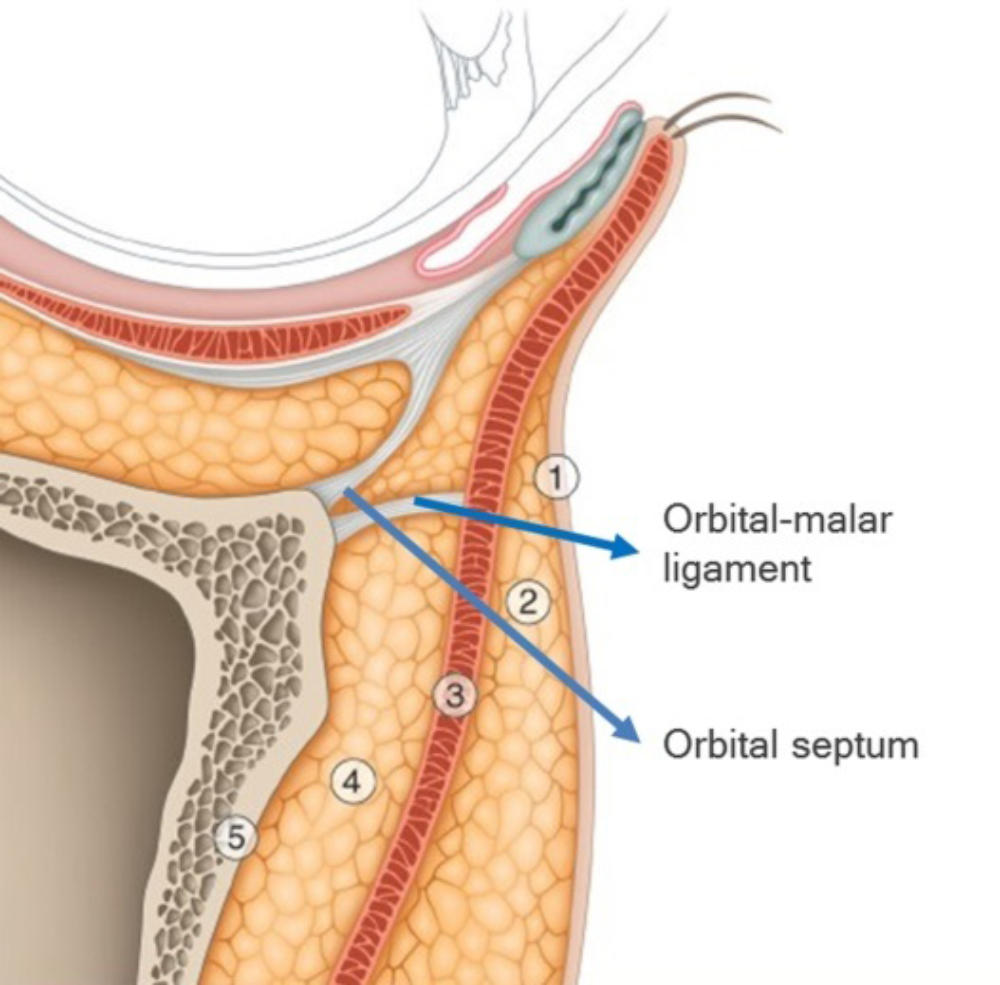
Figure 1: Anatomy of the infraorbital region (courtesy of Teoxane data source).
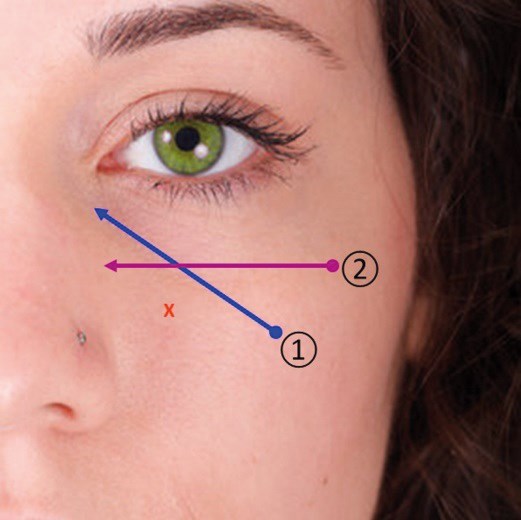
Figure 2: The Two-Point Eye Lift™.
The infraorbital area is particularly vulnerable to early signs of ageing. Protuberance of the lower eyelid occurs primarily due to age-related weakness of the orbital septum, accompanied by pseudoherniation of inferior orbital fat. Thinning of the skin, bone resorption, muscle atrophy, and loss of fatty tissue are also seen with ageing and lead to the formation of the tear trough and palpebremalar groove in infraorbital hollows. Additionally, loss of volume in the mid face, alongside inferior translocation of the malar fat pad leads to increased exposure of the inferior orbital rim. This results in a relative surface depression and apparent increase in lower eyelid length [2].
Procedure
Clinicians undertaking infraorbital rejuvenation with HA dermal fillers should have considerable experience with injectable fillers and be familiar with the specific biochemical specifications as well as clinical characteristics of the HA used. Care should be taken to assess individual patient characteristics, with physical examination and history used to detect signs of fluid retention and possible localised impairment of lymphatic drainage. Pre-treatment photography is essential.
Prior to the procedure, the patient should be positioned with their head firmly against the headrest of a chair with the backrest reclined 30° from the upright position. To assess there is no excessive palpebral laxity, I make sure to perform a ‘snap test’; I also mark out the upper and lower borders of the tear trough and actively palpate and mark out the infraorbital rim. For satisfactory results, some patients benefit from concurrent volume replacement in the mid-face.
To achieve a homogenous placement of the filler, I tend to use a blunt-tipped cannula (specifically 27G or 25G).
Two-Point Eye Lift™ requires only two entry points, and with the use of blunt-tip cannula, this technique minimises the risk of bruising. The filler should be placed just above the periosteum, beneath the OOM, along the orientation of the tear trough (number 1 on Figure 2). To help soften the transition between orbital fat and malar fat, small aliquots of filler are precisely injected along and immediately inferior to the infraorbital rim (number 2 on Figure 2).
To avoid injecting through the orbital septum, exert mild pressure at the orbital margin using the index finger of the contralateral hand [1]. Where possible, avoid using large volumes of filler in any one location and err towards under-correction with a follow-up appointment scheduled for possible touch-up after four weeks.
Possible adverse events and how to avoid these
Two-Point Eye Lift™ has been constructed to minimise side- effects and complications commonly associated with treatment in this area. After the procedure, ice packs can be applied to induce vasoconstriction. Although bruising is not common, the use of a 27G blunt-tipped cannula is capable of lacerating blood vessels if excessive force is used. Choosing non-particulate HA dermal filler of lower viscosity lessens the risk of surface irregularity [3]. Deep tissue massage should be avoided for three weeks to avoid displacement of the filler. The ‘Tyndall effect’ may occur with a superficial placement of HA and is commonly observed when particulate dermal fillers are used in this region. Infection is possible, although the risk can be minimised through the judicious use of antiseptic prior to the treatment and observing a strict protocol for infection control [5]. Product migration can occur in this area; this can be caused by migration of the product from the deep plane through the dissociation of the OOM. Although rare, iatrogenic central retinal artery occlusion secondary to intravascular injection of HA filler has been reported.
Conclusion
The Two-Point Eye Lift™ is an effective and safe technique in infraorbital rejuvenation with HA dermal fillers. It encompasses rejuvenation of both the tear trough and the infraorbital hollows. Risks of side-effects and complications can be minimised by a comprehensive understanding of the anatomy in this region and correct choice of products.
References
1. Finn JC. Fillers in the Periorbital Complex. Facial Plast Surg Clin North Am 2007;15:123-32.
2. Viana GA, Osaki MH, Cariello AJ, et al. Treatment of the tear trough deformity with hyaluronic acid. Aesthet Surg J 2011;31(2):225-31.
3. Goldberg RA, Fiaschetti D. Filling the periorbital hollows with hyaluronic acid gel: initial experience with 244 injections. Ophthal Plast Recons Surg 2006;22:335-43.
4. Rohrich R. Compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg 2007;119:2219-27.
5. Chabra I. Severe site reaction after injecting hyaluronic acid based soft tissue filler. Cosmetic Dermatology 2011;24(1):14-21.
Declaration of competing interests: Dr Bong is the registered trademark owner of Two-Point Eye Lift (TM).
COMMENTS ARE WELCOME





